A Study on Thermal Behavior and Degradation kinetic of Microgels Based on PEG/Poly (NIPAM-co-AMPS)
Ajoy Kumar Saikia*
DOI: 10.22607/IJACS.2022.1001001
Volume 10, Issue 1 | Pages: 1-7
Abstract
Temperature-responsive copolymeric microgels were synthesized via free radical polymerization of N-isopropylacrylamide
(NIPAM) and acrylamido-2-methylpropyl sulphonic acid (AMPS) in presence of PEG as microinitiator under emulsifier free
conditions. The thermal behavior of microgels in water was studied by dynamic light scattering (DLS) at different temperature.
The temperature-dependent equilibrium constant derived from swelling data obtained from DLS and thermodynamic quantities
are measured by Van’t Hoff analysis. The thermogravimetric analysis was used for the evaluation of thermal stability and
degradation kinetics of different microgel samples. The integral procedure decomposition temperature (IPDT) used to estimate
the inherent thermal stability of microgels including volatile parts. The IPDT of microgel sample without AMPS was 368.99°C
and the IPDT increased with increasing AMPS concentration in the feed composition. Thus, the thermal stability increased with
increasing the AMPS concentration in the samples. The activation energy (Ea) of different microgel samples were calculated
by using Broido and Horowitz & Metzger methods. The activation energy was found to be increases from 27.36 KJ/mol to
53.66 KJ/mol depending upon AMPS concentration in the feed composition. The glass transition temperature (Tg) of the
microgel samples were examined by differential scanning calorimetric analysis. The stability and thermal degradation kinetics
are important for designing copolymer blends in different area of applications.
Keywords
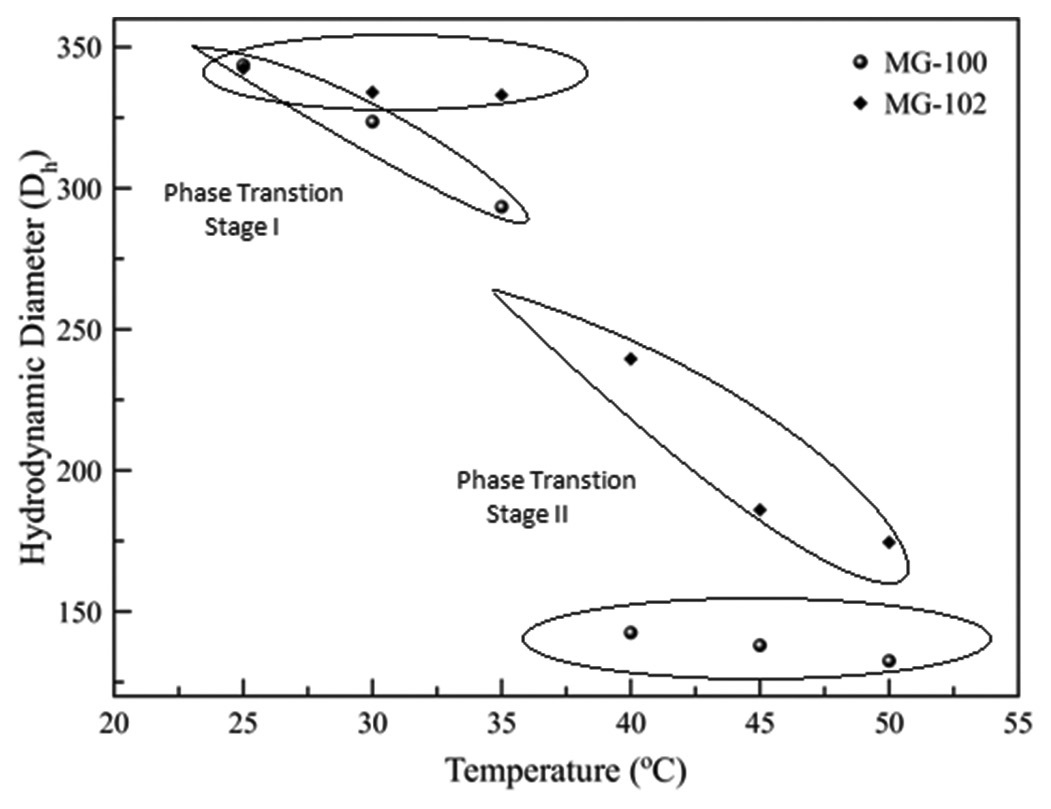
Microgels Thermal degradation Kinetic parameters Integral procedure Decomposition temperature Activation energy. Indian Journal of Advances in Chemical Science Article Indian Journal of Advances in Chemical Science2022; 10(1): 1-7 KROS Publications 2 www.ijacskros.com gravimetric analysis (TGA) and DSC analysis were used for this study. The Broido and Horowitz-Metzger approximate methods are used to calculate the activation energies for comparison. 2. EXPERIMENTAL 2.1. Materials The monomer NIPAM acrylamido-2-methylpropyl sulphonic acid (AMPS) was purchased from Sigma-Aldrich and were used as received PEG-6000 Merck Specilities Pvt. Ltd. Mumbai N N’- methylenebisacrylamide (MBA) ammonium ceric (IV) nitrate 99.99% Sigma-Aldrich was dried in oven at 105°C for 1h prior to use reagent grade nitric acid. 2.2. Preparation of Microgels Polymerization was carried out in a round bottom flask connected to a water condenser 1 g PEG was dissolved in 10 ml of distilled water to this solution 4 ml of 0.1 M (NH4)2Ce(NO3)6 solution prepared in 1N nitric acid was added and stirred for 5 min. Then the measured amount of NIPAM acrylamido-2-methylpropyl sulphonic acid (AMPS) and MBA as cross-linker were added. Nitrogen was purged through the solution for 1 h to remove oxygen prior to polymerization. The polymerization was carried out at 50°C for 4 h under nitrogen atmosphere. The solution was then cooled to room temperature and then centrifuged 0.5 h (10.000 rpm Eppendrof 5810R) at 40°C and then redispersed in distilled water. This process was repeated 3 times and then the samples were dried to a constant weight. 2.3. Characterization of Microgels The hydrodynamic particle size and size distribution of microgel particles were measured at different temperature ranging from 25 to 50°C at constant copolymer concentrations in pure water by dynamic light scattering (DLS) Zetasizer Nano-S Malvern Instrument. The measurement was made at the scattering angle θ =173° wavelength (λ) = 633 nm. The time average scattering intensity correlation functions were obtained with an acquisition time of 30 S for each run. The results of size distribution were analyzed with a Laplace inversion program (CONTIN). Thermal degradation of gels was investigated by a thermogravimetric analyzer (Model Perkin Elmer Pyris Dimond TGA/DTA) from room temperature to 750°C with heating rate 10°C/ min. under nitrogen atmosphere. DSC measurements were performed on a TA instruments DSC Model 2010 in nitrogen atmosphere with 2–5 mg of polymer samples weighed in aluminum pans at a heating rate of 10°C/min. 3. RESULTS AND DISCUSSION 3.1. Thermal Behavior in Water Thermo-responsive microrogels were prepared by free-radical copolymerization of NIPAM and AMPS using PEG as macroinitiator in presence of MBA as cross-linking agent in water at 50°C. During the reaction PEG macroradicals were formed upon redox reaction of the –CH2OH end groups with Ce(IV) ([Ce(IV)]/[OH end group] 1.2/1) in acidic medium [30]. Macroradical initiates water-soluble AMPS MBA and NIPAM monomers which then grow in solution until they reach a critical chain length to become colloidal precursor particles. Either the precursor particles deposit onto an existing polymer particle or aggregate with other precursor particles until they form a particle sufficiently large to be colloidally stable. PEG also helps to increase the colloidal stability of precursor particle. These stable particles create nuclei for growing polymer chains indicated by turning of clear reaction mixture into milky. The formation of particles through precipitation polymerization mechanism is called soap-free emulsion polymerization. Due to the presence of negatively charged AMPS comonomer in the polymerization system there is strong electrostatic repulsion among the sulfonate anion (-SO3-) which results in the expansion of network structure of microgels and have high water absorption properties. The characterizations of particles size and particles size distribution of temperature-responsive microgels were investigated by means of DLS at different temperatures was reported in our previous article [40]. The swelling curves obtained show two-step process where gel particles shift from swollen to collapsed state at temperature range 30–40°C as shown in Figure 1. The phase transition is an equilibrium process of the competing swollen and collapsed state. The equilibrium constant Keq which is equal to the ratio of (Collapsed)/(Swollen) [41] and determined from the swelling data of microgels. The cloud point measurement or DSC measurement done extensively used for the determination of phase transition of temperature of microgels [37-39]. Here van’t Hoff equation is used to measure ΔH ΔS ΔG of microgels sample from van’t Hoff plot obtained by plotting lnKeq versus 1/T which show two-stage process with different enthalpy entropy component. The slope and intercept of the straight line give ΔH and ΔS respectively as shown in Figure 2. The enthalpy for stage I is endothermic i.e 6.29 and 2.13 kJ/mol; entropy –6.53 and –5.32 J/mol K for MG-100 and MG-102 respectively. In stage I microgels show water swelling properties due to the formation of hydrogen bond between water molecules and amide groups of PNIPAM PAMPS and ionization of sulfonic acid group in PAMPS. At the transition temperature hydrogen bonding between the polymer and water molecules disrupted result dehydration of the system and water is released out of the polymer network with a large gain in entropy. The enthalpy for the Stage II is also endothermic i.e. 12.18 and 26.01 kJ/mol; entropy value 4.26 and 5.45 J/mol K for MG-100 & MG-102 respectively. The enthalpy obtained in Stage II is comparable to water hydrogen bond ΔH≈ 10 kJ/mol [42] thus; it assigned the loss of hydrogen bond above the transition temperature of polymer. 3.2. Thermal Stability Thermogravimetric analysis is one of the common techniques to investigate the thermal stability and decomposition of different polymeric materials as a function of temperature or time [43]. Figure 3 shows the TGA thermograms of microgels with different concentrations Figure 1: Hydrodynamic diameter of microgels at different temperature.References
No references available for this article.
Citation
Ajoy Kumar Saikia*. A Study on Thermal Behavior and Degradation kinetic of Microgels Based on PEG/Poly (NIPAM-co-AMPS). Indian J. Adv. Chem. Sci. 2022; 10(1):1-7.