3ʹ-Aminofluorene-9-spiro-5ʹ-imidazolidine-2ʹ,4ʹ-dithione as a Sensing Material for Planar Aromatic Solvents
Tomomi Sakata, Mitsuaki Suzuki
DOI: 10.22607/IJACS.2022.1001005
Volume 10, Issue 1 | Pages: 22- 28
Abstract
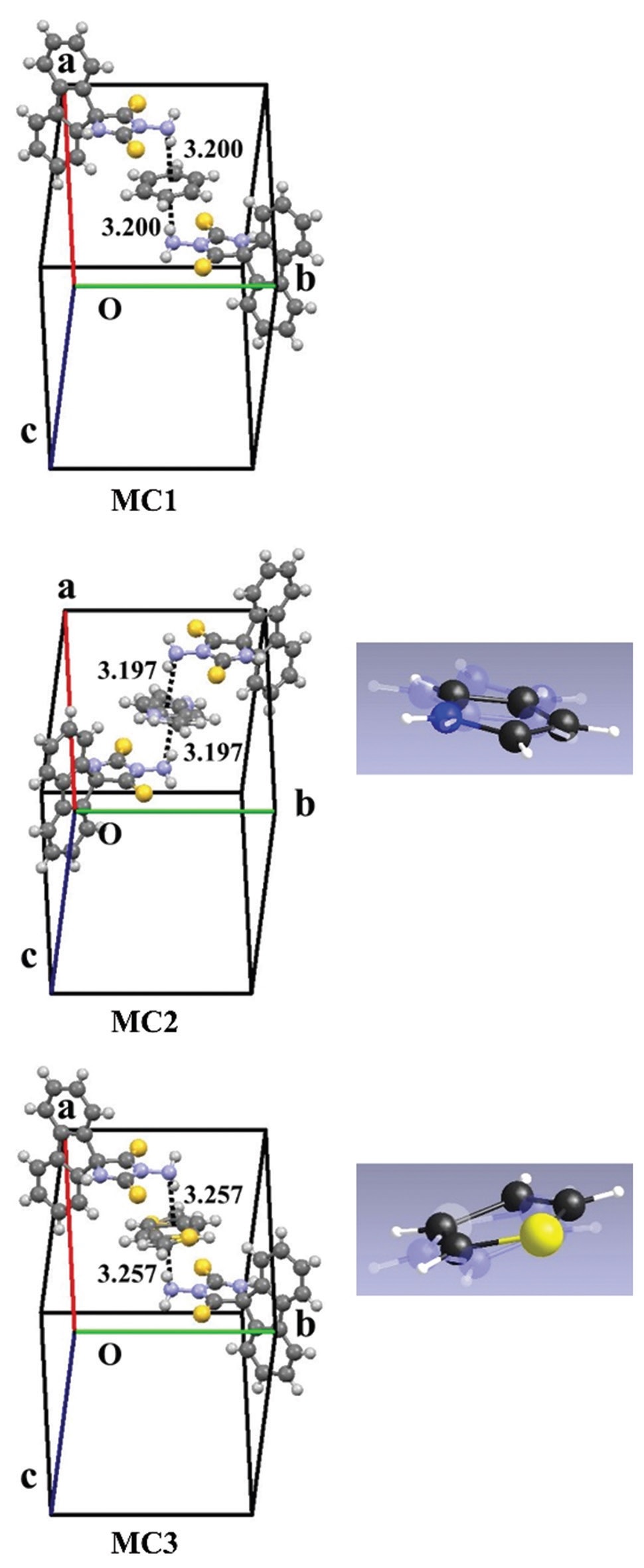
Herein, we investigate the non-covalent interactions between aromatic compounds and a spirocycle-bearing imidazolidinedithione
molecule 3ʹ-aminofluorene-9-spiro-5ʹ-imidazolidine-2ʹ,4ʹ-dithione Host Material (HM). HM and planar aromatic solvent
molecules, such as benzene, pyrrole, and thiophene, form molecular compounds with a composition ratio of 2:1 through NH∙∙∙π
interactions. These compounds exhibit weak emission in the λmax range of 515–544 nm, which were estimated from the sum of the
squares of the differences between the electronegativities of the bonded atoms comprising the guest molecule structures without
employing quantum calculations. Moreover, a linear correlation between the desorption temperature and NH∙∙∙π interactions is
determined from the heats of formation of HM, guest molecules, and corresponding molecular compounds.
Keywords
3ʹ-aminofluorene-9-spiro-5ʹ-imidazolidine-2ʹ 4ʹ-dithione Molecular compound N-H∙∙∙π interaction Planar aromatic solvent.References
No references available for this article.
Citation
Tomomi Sakata, Mitsuaki Suzuki. 3ʹ-Aminofluorene-9-spiro-5ʹ-imidazolidine-2ʹ,4ʹ-dithione as a Sensing Material for Planar Aromatic Solvents. Indian J. Adv. Chem. Sci. 2022; 10(1):22- 28.