Synthesis, Physico-chemical, Spectral, and Antibacterial Activity Studies of Ni(II), Mn(II), and Cu(II) Complexes of Schiff Base Ligand Containing Triazole moiety
Nayaz Ahmed*, Mohd Riaz, Suman Malik, Archana Singh
DOI: 10.22607/IJACS.2022.1002002
Volume 10, Issue 2 | Pages: 59-65
Abstract
A new series of Ni(II), Mn(II), and Cu(II) complexes of tridentate Schiff base derived from condensation of 3-Amino-1,2,4-
triazole (3-Amino-s-Triazole) with salicylaldehyde (2-Hydroxybenzaldehyde) were synthesized in a 2:1 molar ratio. Physical
(Magnetic measurements, molar conductance, and TG), spectral (IR, UV-Vis, and Mass) and analytical data have established the
structures of synthesized Schiff base and its metal complexes. The elemental analysis data suggest the stoichiometry to be 2:1
(L: M). All the complexes are non-electrolytic in nature as suggested by molar conductance measurement. Infra-red spectral data
indicate the coordination between the ligand and the central metal ion through deprotonated phenolic oxygen, triazole nitrogen,
and azomethine nitrogen atoms. Spectral studies and magnetic susceptibility measurements suggest an octahedral geometry
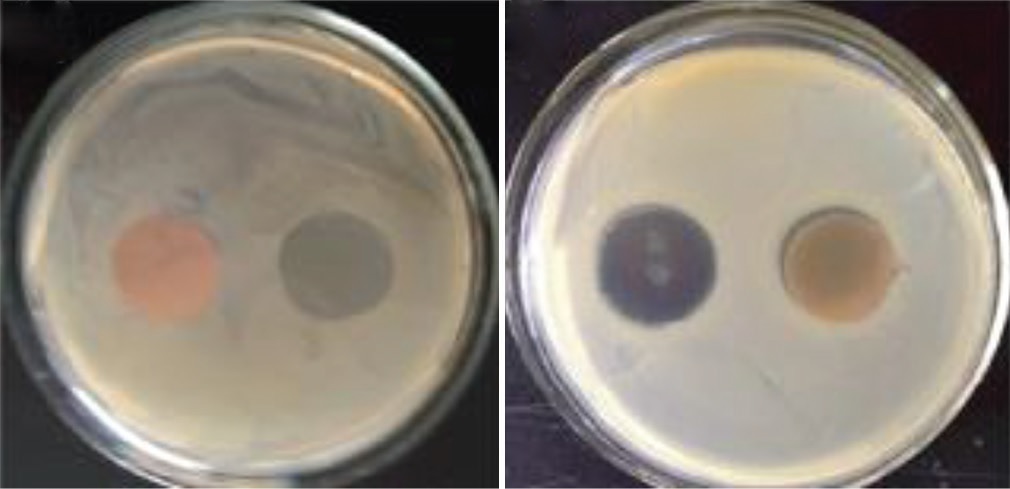
for the complexes. The ligand and its complexes were tested/screened for their antibacterial activity against two Gram-positive
bacteria species Staphylococcus aureus and Lactobacillus and two Gram-negative species Escherichia coli and Pseudomonas
aeruginosa. It is observed that the ligand as well as the complexes showed good activity against all microbes.
Keywords
3-Amino-1 2 4-triazole Antibacterial activity Complexes Schiff base Spectral studies.References
No references available for this article.
Citation
Nayaz Ahmed*, Mohd Riaz, Suman Malik, Archana Singh. Synthesis, Physico-chemical, Spectral, and Antibacterial Activity Studies of Ni(II), Mn(II), and Cu(II) Complexes of Schiff Base Ligand Containing Triazole moiety. Indian J. Adv. Chem. Sci. 2022; 10(2):59-65.