Removal of Methylene Blue by Adsorption onto Activated Biomass of Prunus cerasus. Batch Study
A. Kale Anandrao*
DOI: 10.22607/IJACS.2022.1002005
Volume 10, Issue 2 | Pages: 79- 84
Abstract
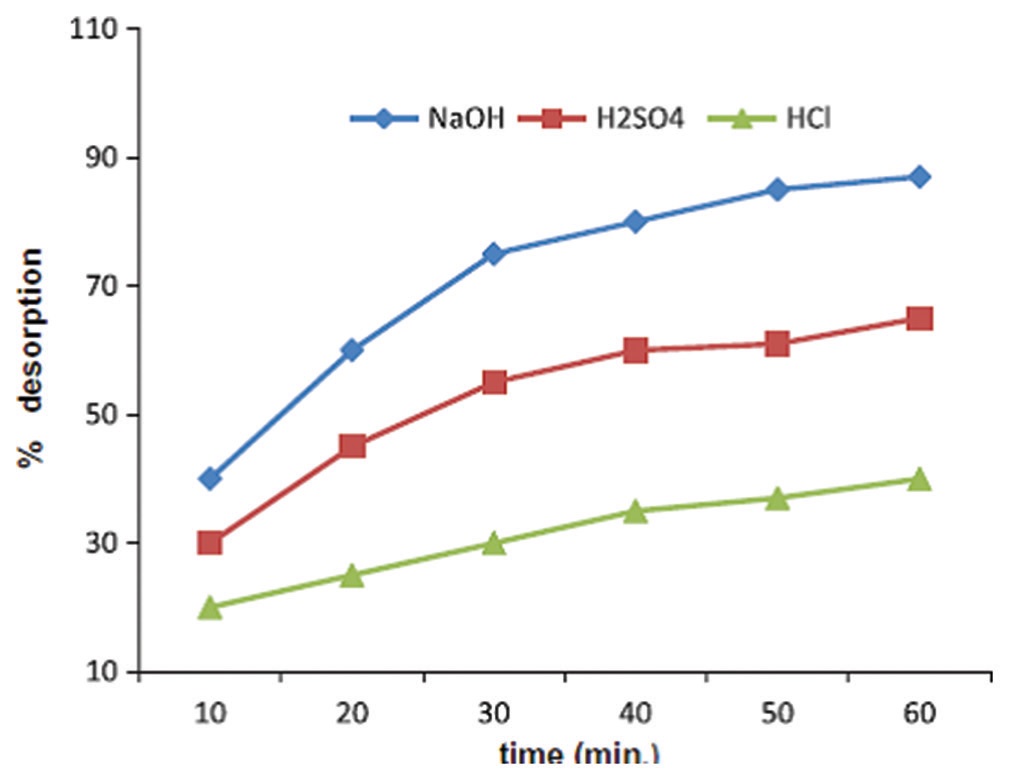
The present study deals with investigation of methylene blue by adsorption onto activated biomass of Prunus cerasus (ACBPC),
the experimental work was carried out on methylene blue (MB) removal by ACBPC. The effect of various factors such as contact
time, adsorbent dosage, initial dye concentration, temperature, and pH of dye solution was investigated. The maximum MB
removal was observed at pH 8. The adsorption equilibrium was represented with Langmuir, Freundlich, and Temkin isotherm
models. Langmuir and Tempkin equations were found to have the correlation coefficient value in good agreement. The high
value of correlation coefficient 0.998 indicates the applicability of Langmuir isotherm which assumes a monolayer coverage
and uniform activity distribution on the sorbent surface. Adsorption of MB onto ACBPC followed pseudo second order kinetics.
The calculated values of DH°, DS°, and DG° were found to be 19.30 kJ/mol, 73.35 kJ/mol, and –1.54 kJ/mol, respectably.
Adsorption process was spontaneous and endothermic in nature.
Keywords
Activated biomass of Prunus cerasus Adsorption isotherm Methylene blue.References
No references available for this article.
Citation
A. Kale Anandrao*. Removal of Methylene Blue by Adsorption onto Activated Biomass of Prunus cerasus. Batch Study. Indian J. Adv. Chem. Sci. 2022; 10(2):79- 84.