Extraction Equilibria of Cobalt (II) with 3-Hexylaminopyridine in Kerosene from Aqueous Solution in Chloride Media
Maroti V. Kanetkar*, Jivan M. Kondare, Gopal K. Kakade
DOI: 10.22607/IJACS.2022.1003001
Volume 10, Issue 3 | Pages: 100-103
Abstract
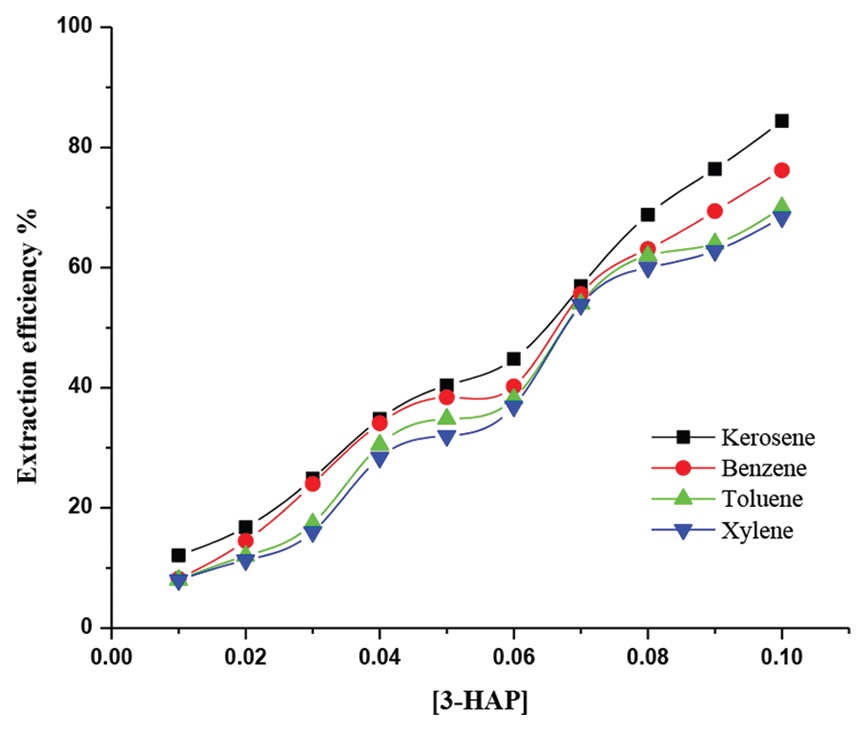
Extraction equilibria of cobalt (II) from aqueous chloride solutions with 3-hexylaminopyridine (3-HAP) dissolved in kerosene
was investigated. Results showed that KCl salt can greatly improve the cobalt (II) extraction efficiency. The distribution
coefficients were significantly dependent on the concentration of chloride ions and the pH value in the aqueous phase because
of mechanism of extraction and complex formation in the organic phase. With initial pH value of pH 1–2 and chloride ion
concentration 1.0 mol L−1, the maximum extraction (>90%) was observed. In addition, 2.0 mol L−1–2.5 mol L−1 concentrations
of hydrochloric acid in aqueous phase increase the extraction efficiency. Extraction equilibrium by 3-HAP with cobalt (II) was
established within 10 min.
Keywords
3-Hexylaminopyridine Extraction equilibria Cobalt (II) KeroseneReferences
No references available for this article.
Citation
Maroti V. Kanetkar*, Jivan M. Kondare, Gopal K. Kakade. Extraction Equilibria of Cobalt (II) with 3-Hexylaminopyridine in Kerosene from Aqueous Solution in Chloride Media. Indian J. Adv. Chem. Sci. 2022; 10(3):100-103.