Effect of HNO3 on the Extraction of Uranium (VI) using 1-Phenyl-3-methyl-4- trichloroacetyl pyrazolone-5 (HTCP) at Different Acid Concentrations
Victoria Bennett*, Nimibofa Ayawei
DOI: 10.22607/IJACS.2022.1003002
Volume 10, Issue 3 | Pages: 104-108
Abstract
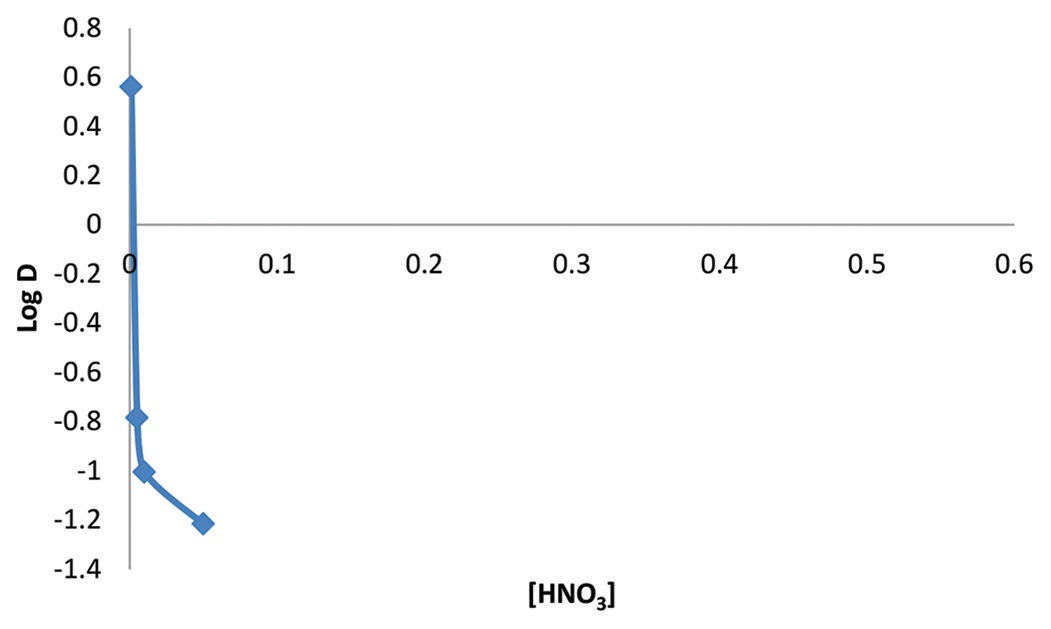
The effect of HNO3 in the extraction U (IV) from buffered aqueous solution using a chloroform solution of the ligand 1-phenyl-
3-methyl-4-trichloroacetyl pyrazolone-5 (HTCP) was studied. This was carried out using solvent-solvent extraction, with 0.001
M, 0.005 M, 0.01 M, 0.05 M, 0.1 M, and 0.5 M aqueous phases of HNO3. The working concentration was 200 mg/L. The
extraction was carried out using a chloroform solution of 0.05 M (HTCP). The solution was agitated with an equilibration
time of 30 min, after which the phases were allowed to settle, and then, the aqueous raffinate was withdrawn and analyzed by
difference colorimetrically using 1,10-phenanthroline. Distribution ratios (D) and percentage extractions (% E) were calculated
by difference. The results obtained showed that the interference of nitrate ions was more pronounced as nitric acid concentration
got >0.01 M HNO3. U(VI) was completely masked at concentrations greater than 0.01 M, indicating that HNO3 is only a good
extraction medium for the extraction of U(VI) at lower concentrations. There was optimum % extraction of 95.59 %, 78.41%,
and 33.92 % at molar concentrations 0.001 M, 0.01 M, and 1 M, respectively. An adduct complex of the metal characterized
as UO2 (NO3) was extracted. Complete recovery of U(VI) will require two or three batches of extraction using the acid at
concentration 0.001 M where U(VI) was best extracted.
Keywords
Concentration Extraction HNO3 Interference MaskingReferences
No references available for this article.
Citation
Victoria Bennett*, Nimibofa Ayawei. Effect of HNO3 on the Extraction of Uranium (VI) using 1-Phenyl-3-methyl-4- trichloroacetyl pyrazolone-5 (HTCP) at Different Acid Concentrations. Indian J. Adv. Chem. Sci. 2022; 10(3):104-108.