Synthesis and Characterization of Boron Derivatives with N-(o-hydroxy substituted benzyl)phenylalanines
Anil Kumar*, Manju Kandpal, Supriya Verma, Seema Islam, Pradeep Bhatt
DOI: 10.22607/IJACS.2022.1003010
Volume 10, Issue 3 | Pages: 151-158
Abstract
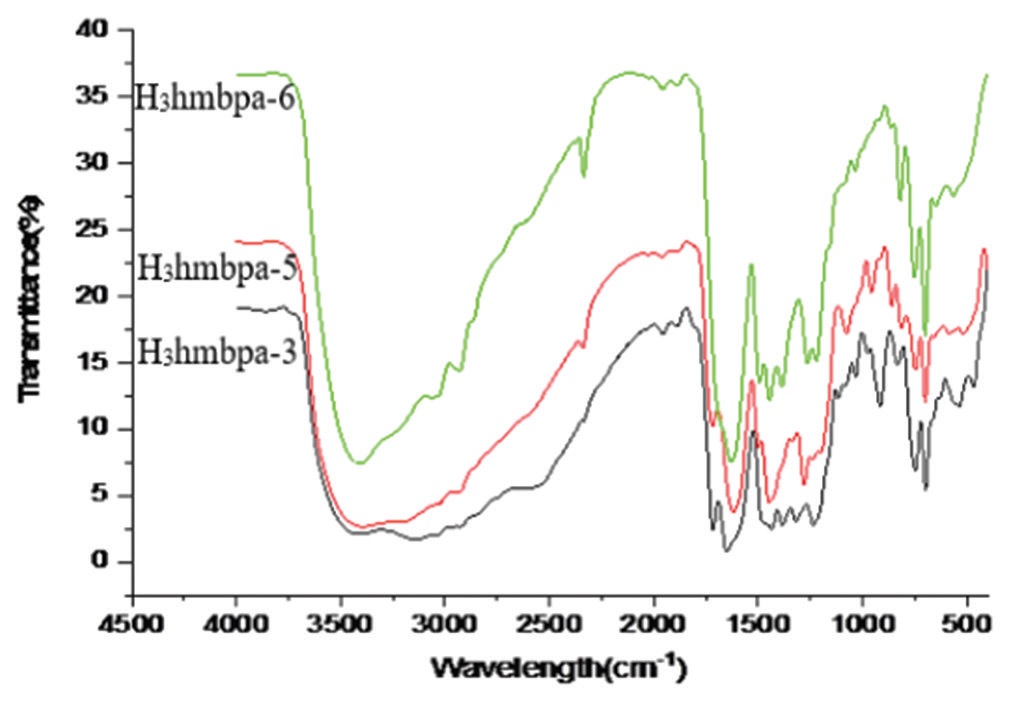
An efficient and general method has been developed for synthesis of boron derivatives of N-(o-hydroxy substituted benzyl)
phenylalanines from boron triisopropoxide and protonated ligands H3hmbpa-3[N-(2-hydroxy-3-methyl benzyl)phenylalanine],
H3hmbpa-6[N-(2-hydroxy-6-methyl benzyl)phenylalanine], and H3hmbpa-5[N-(2-hydroxy-5-methyl benzyl)phenylalanine] in
ratio of 1:1, 1:2, and 1:3 using a highly vacuumed assembly. Boron triisopropoxide was prepared using boric acid and isopropyl
alcohol and the protonated ligands H3hmbpa-3, H3hmbpa-6, and H3hmbpa-5 were prepared from reaction between o,m,p-
cresol, formaldehyde, and phenylalanine. The structure of boron triisopropoxide, ligands, and boron derivatives is identified
by H1NMR, FT-IR, and elemental analysis. The elemental analysis, H1NMR, and FT-IR data of boron derivatives proposed the
boron: Ligands stoichiometry, molecular formulae, and coordination linkage of ligands with boron in 1:1, 1:2, and 1:3 ratio. The
number of moles of isopropanol liberated during the preparation of boron derivatives in the binary azeotrope with benzene was
estimated by the method described by Bradey et al. and Mehrotra.
Keywords
Azeotrope Boron triisopropoxide H3hmbpa-3 6 5 O m p-cresol Phenylalanine Stretching.References
No references available for this article.
Citation
Anil Kumar*, Manju Kandpal, Supriya Verma, Seema Islam, Pradeep Bhatt. Synthesis and Characterization of Boron Derivatives with N-(o-hydroxy substituted benzyl)phenylalanines. Indian J. Adv. Chem. Sci. 2022; 10(3):151-158.