2-Pyrrolidin-2yl-1H-benzimidazole (PBI): An efficient organocatalyzed synthesis of β-sulphido carbonyl compounds
Gaurav Praksh, Ajay Kumar Das
DOI: 10.22607/IJACS.2022.1004007
Volume 10, Issue 4 | Pages: 184-188
Abstract
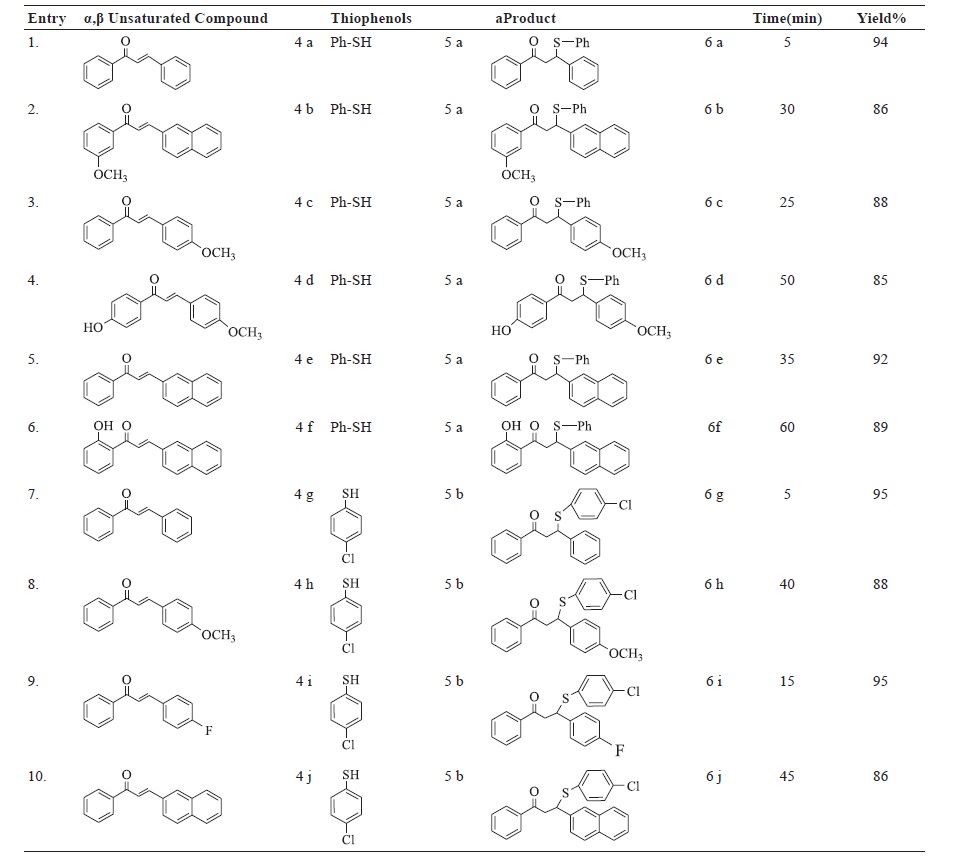
An efficient and organocatalytic synthetic protocol has been developed involving 2-Pyrrolidin-2yl-1H-benzimidazole (PBI)
catalyzed synthesis of β-sulfido carbonyl compounds through reaction of thiophenols with α,β-unsaturated carbonyl compounds
in good and excellent yield under mild reaction conditions. The synthetic process for PBI organocatalyst has also been developed.
The synthesized organocatalyst was further employed for the synthesis of β-sulfido carbonyl compounds using α,β-unsaturated
carbonyl compounds as substrates. We investigated the appropriated reaction condition by screening different solvents and
conditions. The optimized reaction conditions further utilized to generate the library of β-sulfido analogues. All the synthesized
analogs were characterized using spectroscopic and spectrometric techniques.
Keywords
2-Pyrrolidin-2yl-1H-benzimidazole Conjugate addition Thio-Michael reaction Organocatalysis.References
No references available for this article.
Citation
Gaurav Praksh, Ajay Kumar Das. 2-Pyrrolidin-2yl-1H-benzimidazole (PBI): An efficient organocatalyzed synthesis of β-sulphido carbonyl compounds. Indian J. Adv. Chem. Sci. 2023; 10(4):184-188.