Synthesis, Biological Evaluation, and Docking Studies of 1,2,4,5-Tetrasubstituted Imidazole Derivatives as Antibacterial Agents: Use of Niobia Supported Heteropoly Tungstate as an Efficient Reusable Catalyst
Karunasree Merugu, Aravind Kurnool, Shravya Pachipulusu
DOI: 10.22607/IJACS.2021.901004
Volume 9, Issue 1 | Pages: 22-29
Abstract
The preparation of new 1,2,4,5-tetrasubstituted imidazoles was carried out in a single molecular motif using niobia supported
heteropoly tungstate as an effective catalyst. The efficient condensation of benzil, 3-aryl-1-phenyl-1H-pyrazole-4-carbaldehyde,
aromatic amine, and ammonium acetate was achieved using heteropoly tungstate supported on niobia under both the conventional
and non-conventional conditions. The employed protocol provides significant advantages, as it exhibits a remarkable catalytic
activity on recovery, excellent yields, and excellent reaction efficacy within short reaction times between 1–2 h (conventional)
and 1–3 min (MWI). The structures of all the synthesized products were established by means of spectral and elemental analysis
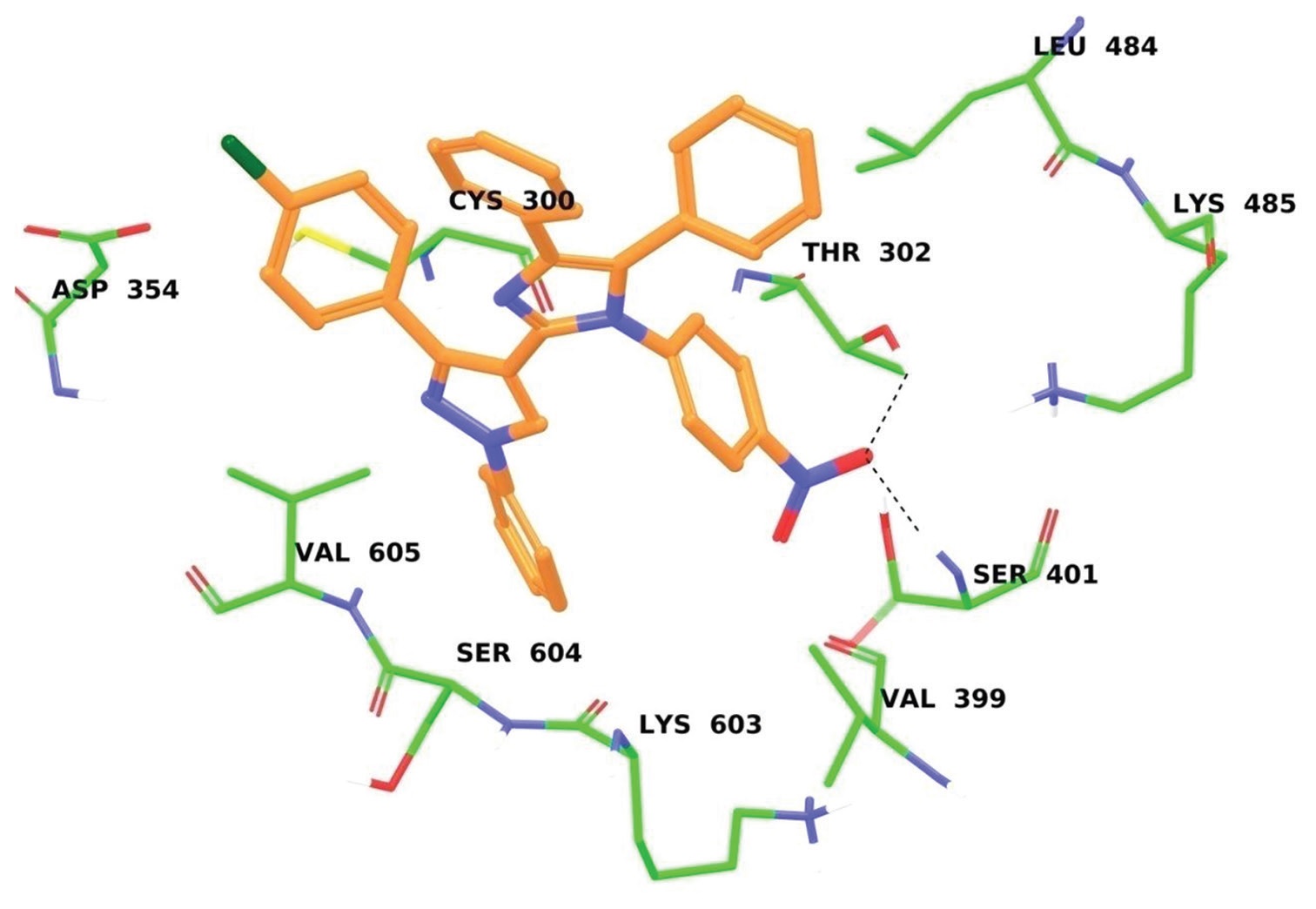
data. They were also screened for their antibacterial activity. To predict the binding mode of compounds with glutamine-fructose-
6-phosphate transaminase (GlcN6P synthase), docking studies were performed.
Keywords
1 2 4 5-Tetrasubstituted imidazoles Single molecular motif Niobia tungstophosphoric acid Microwave irradiation Antibacterial agents.References
No references available for this article.
Citation
Karunasree Merugu, Aravind Kurnool, Shravya Pachipulusu. Synthesis, Biological Evaluation, and Docking Studies of 1,2,4,5-Tetrasubstituted Imidazole Derivatives as Antibacterial Agents: Use of Niobia Supported Heteropoly Tungstate as an Efficient Reusable Catalyst. Indian J. Adv. Chem. Sci. 2021; 9(1):22-29.