Adsorption of Remazol Black 5 and Indigo Carmine on Corn Cobs Activated Carbon: Kinetic, Equilibrium, and Thermodynamic Studies
Abo Essis Armand, Kouakou Yao Urbain*, Yobouet Yao Augustin, Trokourey Albert
DOI: 10.22607/IJACS.2021.902004
Volume 9, Issue 2 | Pages: 69-75
Abstract
In the present work, the study of the adsorption of Indigo Carmine (IC) and Remazol black 5 (RB5) dyes on activated carbon
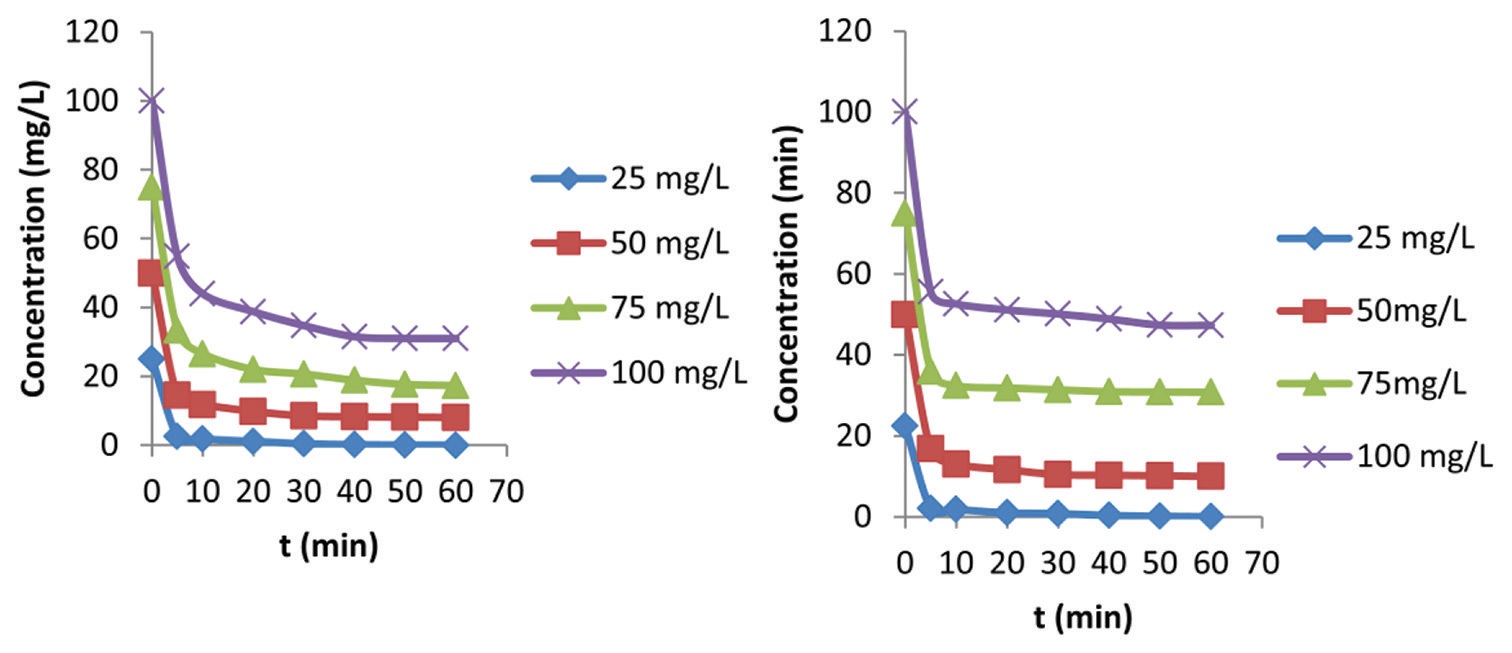
produced from corn cobs (corn cob activated carbon [CCAC]) was performed. The adsorption kinetics of each dye followed a
pseudo-second-order kinetic model with high correlation coefficients (R2 > 0.99) with 30 min as equilibrium time. Parameters
such as the initial dye concentration, the initial pH of the solution and the temperature were well investigated. The amount of
dyes adsorbed increases with the initial concentration and decreases with the increase of pH. The optimal pH is therefore 2. For
an initial concentration of 25 mg/L, the rate of elimination at pH = 2 is 99.96% and 96.92%, respectively, for IC and RB5 at 25°C.
The adsorption of these dyes also increases with temperature. With an initial concentration of 100 mg/L, the percentage dye
uptake increased from 62.14% at 25°C to 99.99 % at 70°C for IC and from 47.12% at 25°C to 99.93 % at 70°C for RB5. These
adsorption processes are spontaneous and endothermic. The Freundlich and Langmuir models well describe the phenomenon of
adsorption which is physisorption in nature. When IC and RB5 are mixed, the adsorption capacity of CCAC decreases.
Keywords
Corn cobs activated carbon Adsorption Thermodynamics Kinetics Indigo carmine Remazol black 5.References
No references available for this article.
Citation
Abo Essis Armand, Kouakou Yao Urbain*, Yobouet Yao Augustin, Trokourey Albert. Adsorption of Remazol Black 5 and Indigo Carmine on Corn Cobs Activated Carbon: Kinetic, Equilibrium, and Thermodynamic Studies. Indian J. Adv. Chem. Sci. 2021; 9(2):69-75.