Physicochemical Investigation of Diverse Interactions of Some Biologically Potent Molecules Prevalent in Aqueous Ionic Liquid Solutions at Different Temperatures
Sukdev Majumder, Kanak Roy, Sanjoy Barman, Mahendra Nath Roy*
DOI: 10.22607/IJACS.2021.902007
Volume 9, Issue 2 | Pages: 89-97
Abstract
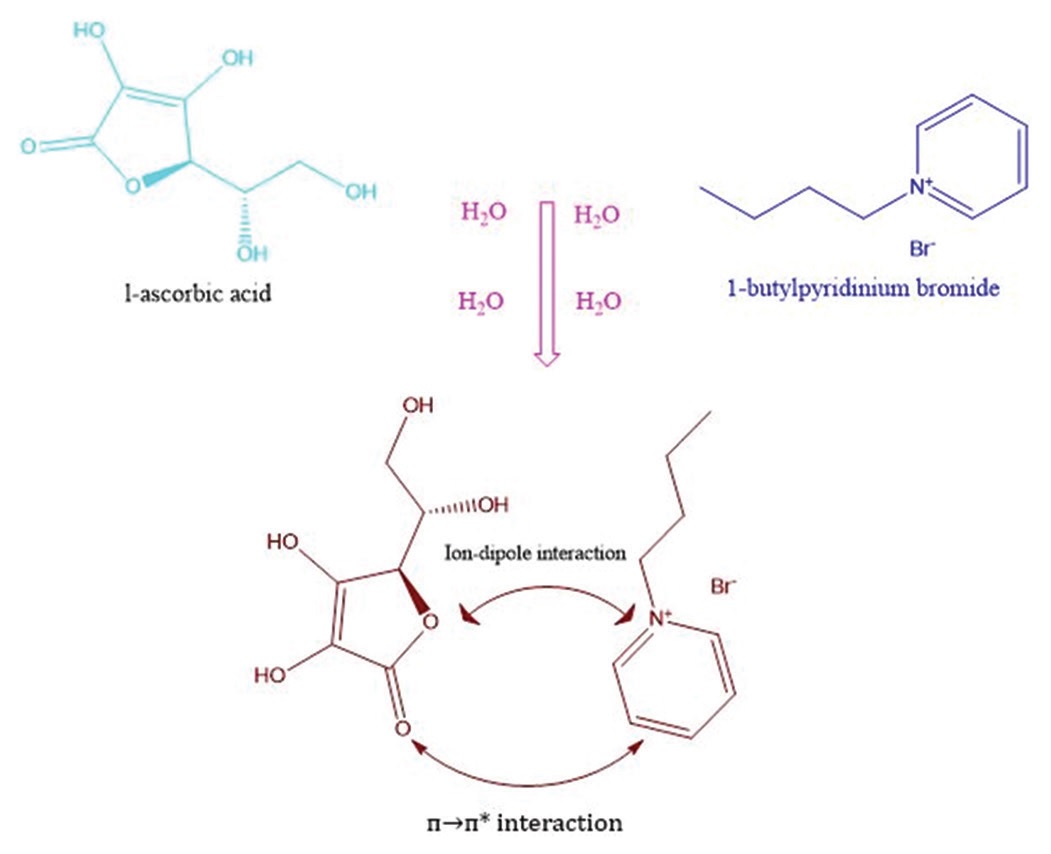
Densities, viscosities, conductivity, and refractive indices of L-ascorbic acid in aqueous solutions of an ionic liquid,
1-butylpyridinium bromide, have been measured at five different temperatures ranging from 293.15K to 313.15K. Some
important parameters have been derived from the above physicochemical experiments, namely, limiting apparent molar volume
(φV0) and viscosity B-coefficients using extended Masson equation and Jones-Dole equation, respectively. Lorentz-Lorenz
equation has used to evaluate molar refractive index (RM) and limiting molar index (RM0). Specific conductivity measurement
applied to ascertain ionic nature of the system.
Keywords
Solute-co-solute interactions Apparent molar volume Viscosity B-coefficient Molar refraction Conductivity studyReferences
No references available for this article.
Citation
Sukdev Majumder, Kanak Roy, Sanjoy Barman, Mahendra Nath Roy*. Physicochemical Investigation of Diverse Interactions of Some Biologically Potent Molecules Prevalent in Aqueous Ionic Liquid Solutions at Different Temperatures. Indian J. Adv. Chem. Sci. 2021; 9(2):89-97.