Spectroscopic Investigation of Steroidal Benzothiazoles as DNA-Binding Agents and their Role in Cytotoxicity
Ayaz Mahmood Dar*, Shafia Mir, Manzoor Ahmad Gatoo, Shamsuzzaman
DOI: 10.22607/IJACS.2021.903001
Volume 9, Issue 3 | Pages: 98-104
Abstract
The new steroidal benzathiazole derivatives (4-6) were synthesized by the reaction of steroidal ketones (1-3) with 2-aminothiophenol
in chloroform. After characterization by spectral and analytical data, the DNA interaction studies of one of the compounds were
carried out by ultraviolet–visible, fluorescence spectroscopy, viscosity measurements, circular dichroism, molecular docking, and
gel electrophoresis. The compound binds to DNA preferentially through electrostatic and hydrophobic interactions with Kb; 4.5
×104 M−1 revealing its potential propensity toward computed tomography DNA. Gel electrophoresis demonstrated that compound 5
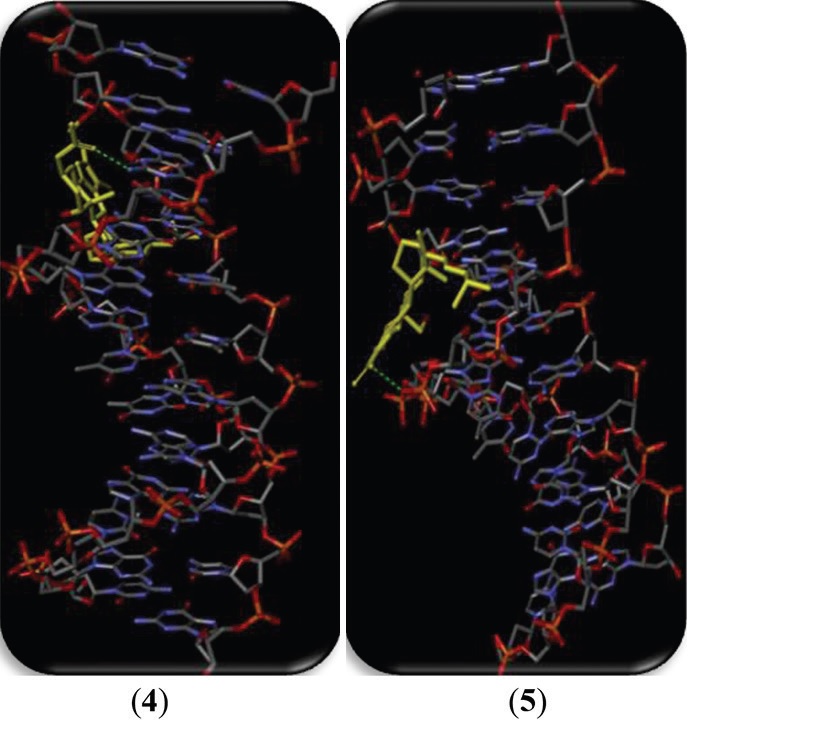
showed a strong interaction during the concentration-dependent cleavage activity with pBR322 DNA. The molecular docking study
suggested the intercalation of compounds in the minor groove of DNA. During in vitro cytotoxicity (3-(4,5-dimethylthizao1-2-y1)-
2,5-diphenyltetrazolium bromide assay), compound (4-6) revealed potential toxicity against the different human cancer cells. Western
blotting analysis clearly indicates that compound 5 causes apoptosis in HL-60 cancer cells. The results revealed that compound 5 has
better prospectus to act as a cancer chemotherapeutic candidate, which warrants further in vivo anticancer investigations.
Keywords
Benzothiazole DNA binding 3-(4 5-Dimethylthizao1-2-y1)-2 5-diphenyltetrazolium bromide assay Western blotting.References
No references available for this article.
Citation
Ayaz Mahmood Dar*, Shafia Mir, Manzoor Ahmad Gatoo, Shamsuzzaman. Spectroscopic Investigation of Steroidal Benzothiazoles as DNA-Binding Agents and their Role in Cytotoxicity. Indian J. Adv. Chem. Sci. 2021; 9(3):98-104.