Synthesis and Characterization of Zinc (II) Complex of Schiff Base Derived from p-Chlorobenzaldehyde
M. Kalaimathi*, Y.Yesu Thangam, R. Ebenezer, R. Anitha
DOI: 10.22607/IJACS.2021.903009
Volume 9, Issue 3 | Pages: 158-161
Abstract
Schiff base complex of Zinc (II) ion has been derived from benzoyl acetone, p-chlorobenzaldehyde, and o-anisidine. The ligand,
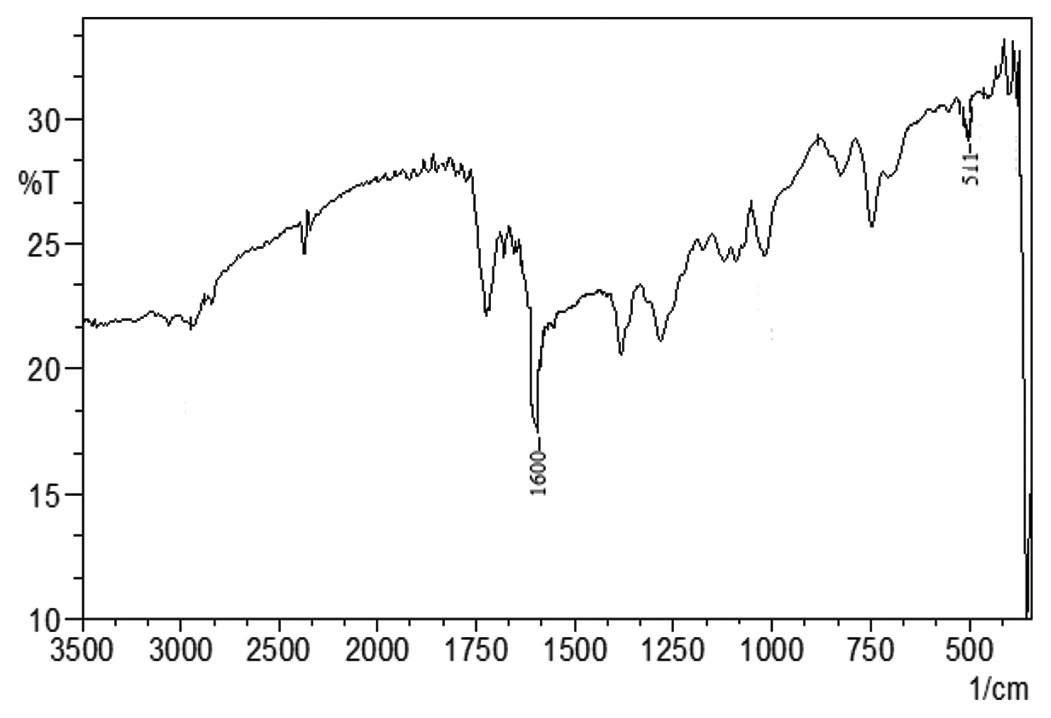
as well as metal complex, was characterized by elemental analysis, UV-visible, magnetic moments, IR, 1H NMR, 13C NMR.
Non-electrolytic nature of zinc (II) complex was proved by the molar conductance data. The antimicrobial activities of the ligand
(L1) and its zinc (II) complex against the bacterial strains such as Escherichia coli, Bacillus subtilis, Staphylococcus aureus and
the fungus Candida albicans, Aspergillus niger, Aspergillus flavus have been carried out. The effect of the substituent on the
biological activity of Schiff base and its complex was investigated. Antibacterial and antifungal activity of the metal complex
is greater than the free ligand (L1) due to chelation and the presence of the electron releasing methoxy group as a substituent in
the aromatic portion of the complex.
Keywords
Schiff base ligand Zinc (II) complex Biological activity Chelation Antimicrobial activity.References
No references available for this article.
Citation
M. Kalaimathi*, Y.Yesu Thangam, R. Ebenezer, R. Anitha. Synthesis and Characterization of Zinc (II) Complex of Schiff Base Derived from p-Chlorobenzaldehyde. Indian J. Adv. Chem. Sci. 2021; 9(3):158-161.