Removal of Rhodamine 6G dye from Water Solution by Alt-maleic anhydride-styrene Copolymer, Cross-linked with Glycerin
Oktay Akperov*, Fatima Kamranzadeh, Elchin Akperov, Mirheydar Abbasov, Leyla Durmazucar
DOI: 10.22607/IJACS.2021.903011
Volume 9, Issue 3 | Pages: 166-173
Abstract
alt–Maleic anhydride–styrene copolymer, crosslinked with glycerin has been used for removal of Rhodamine 6G dye from aqueous
solution. Effects of pH solution, contact time, sorbent dosage, initial concentration of dye and temperature on removal degree have
been evaluated. The equilibrium sorption data were fitted to Langmuir, Freundlich and D–R isotherms. The maximum equilibrium
sorption capacity from the Langmuir equation is equal to 14.73 mg g–1. The found value of the mean sorption energy from D–R
equation (5.472 kJ mol–1) specifies that, sorption of the Rhodamine 6G dye by synthesized polymer–sorbent has a physical character.
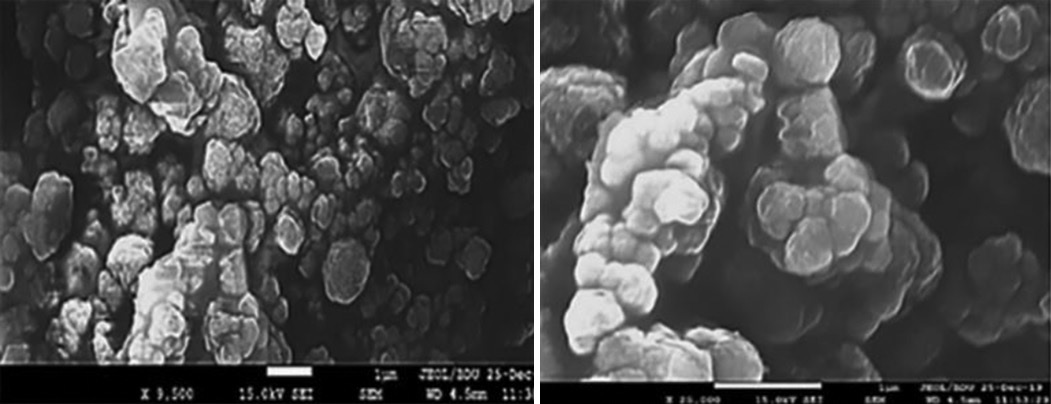
The morphological features and surface characteristics of the dye–loaded sorbent after sorption have been studied using SEM
measurement. The SEM image indicates that the surface of the sorbent non–homogeneous, which connected with accumulation
of the Rhodamine 6G dye molecules on the sorbent surface. The kinetic data were correlated well with the pseudo–second order
model. The positive values of the thermodynamic parameters ΔG° (7.45-6.04 kJ mol–1), ΔH° (18.378 kJ mol–1) and ΔS° (0.0374 kJ
mol–1K–1) indicates that the adsorption process is non–spontaneous, endothermic and randomness in nature.
Keywords
Adsorption Cross-linked copolymer Isotherms Kinetic Rhodamine 6G dye.References
No references available for this article.
Citation
Oktay Akperov*, Fatima Kamranzadeh, Elchin Akperov, Mirheydar Abbasov, Leyla Durmazucar . Removal of Rhodamine 6G dye from Water Solution by Alt-maleic anhydride-styrene Copolymer, Cross-linked with Glycerin. Indian J. Adv. Chem. Sci. 2021; 9(3):166-173.