Non-isothermal Deblocking Studies of Amine Blocked Isocyanates and their Validation Using Numerical Simulation Calculations
Vennila Srinivasan, SumalathaVasam, Sankar Govindarajan*
DOI: 10.22607/IJACS.2021.903015
Volume 9, Issue 3 | Pages: 197-204
Abstract
Influences of isocyanate structure, analytical techniques, and heating rate on deblocking reactions were studied. A series of
N-Methylaniline blocked isocyanates with different isocyanates were synthesized. Their solid and solution state deblocking
reactions were carried out and studied using different analytical techniques as well as different heating rates. The results indicate
that isocyanate structure, the effect of heating rate, analytical method, and state of the blocked isocyanate strongly influence the
deblocking reaction. Solution state deblocking reaction always occurs at lower temperatures compared to solid-state reactions.
Aromatic isocyanate-based blocked isocyanates deblock at lower temperatures compare to aliphatic isocyanate-based blocked
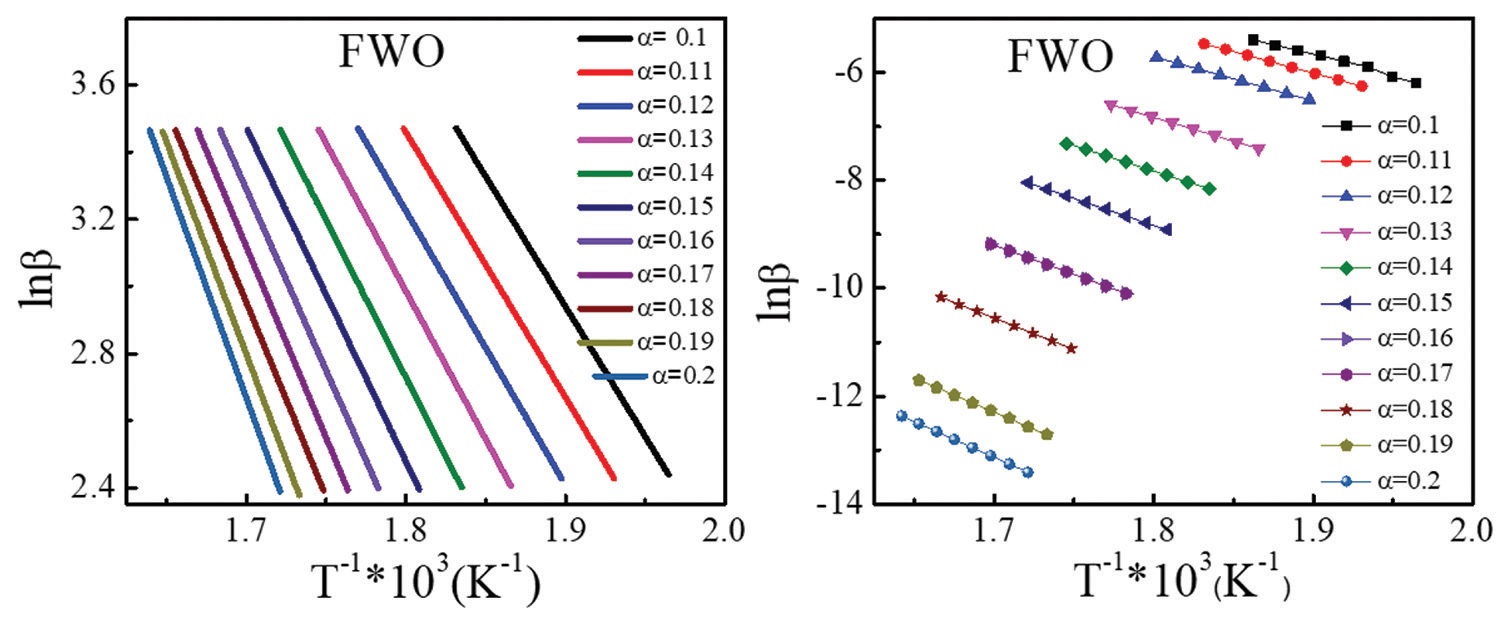
isocyanates. The energy of activation (Ea) for deblocking reaction was determined using conversion degree values obtained from
Thermo-gravimetric analysis and with the help of Friedmann-Reich-Levi and Flynn–Wall–Ozawa equations. Experimentally
determined Ea was validated using numerical simulation calculations.
Keywords
Blocked isocyanates Deblocking Friedmann-Reich-Levi Flynn–Wall–Ozawa Non-isothermal kinetics Simulation.References
No references available for this article.
Citation
Vennila Srinivasan, SumalathaVasam, Sankar Govindarajan*. Non-isothermal Deblocking Studies of Amine Blocked Isocyanates and their Validation Using Numerical Simulation Calculations. Indian J. Adv. Chem. Sci. 2021; 9(3):197-204.