Computational Models: A Sustainable Approach to Reveal the Inhibitory Potential of Benzo(a)anthracene and its Monohydroxy Derivatives Against Human Sex Hormone-Binding Globulin
Nikita Tiwari, Anil Mishra*
DOI: 10.22607/IJACS.2021.904001
Volume 9, Issue 4 | Pages: 250-254
Abstract
Benzo(a)anthracene (BaA), a major environmental pollutant, is metabolized in vivo and produces many hydroxy derivatives.
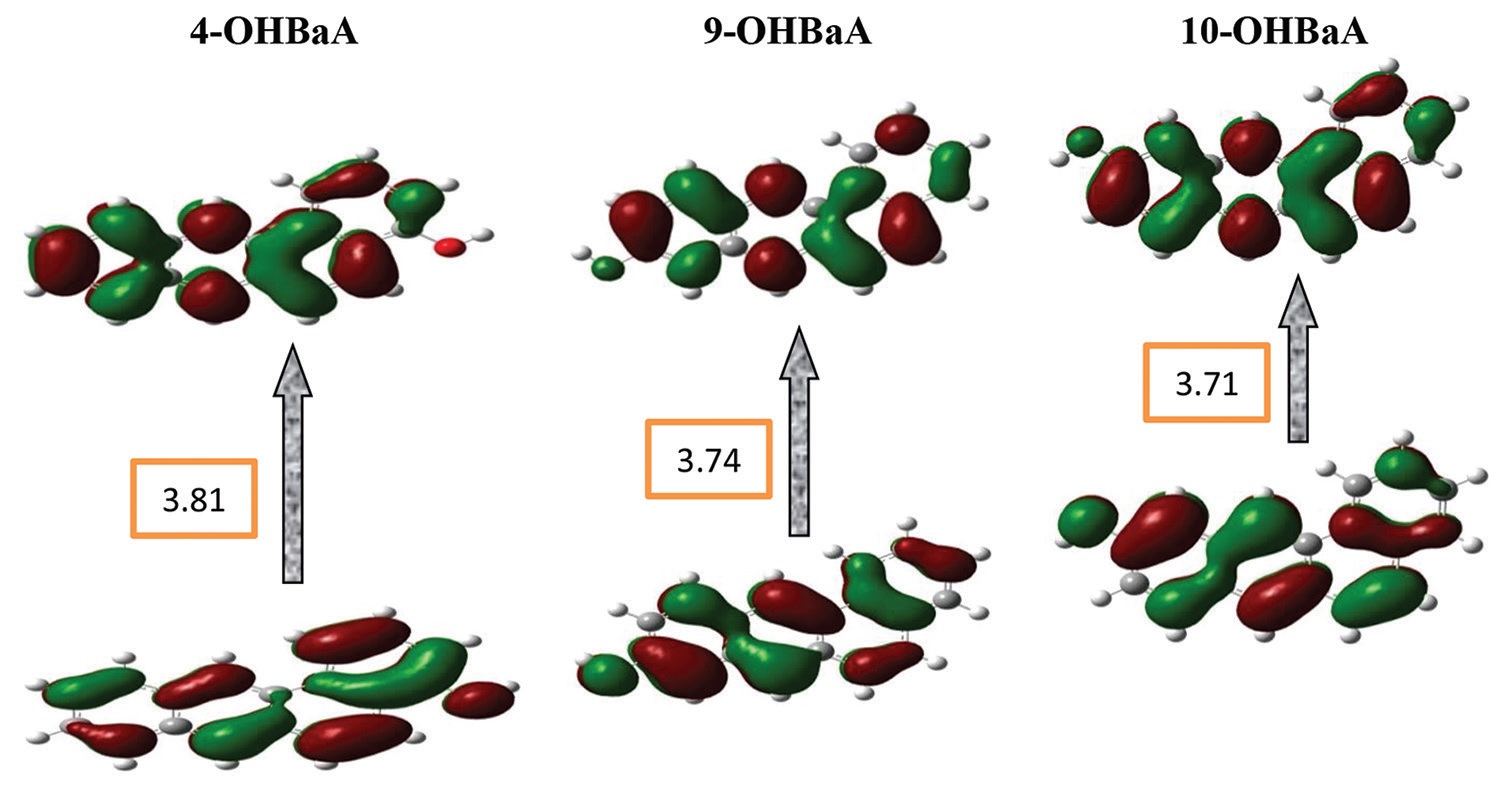
The density functional theory calculations were performed to investigate the frontier molecular orbitals as well as the chemical
reactivity descriptors of BaA and its monohydroxy derivatives using B3LYP/3-21G basis set. Using in silico tools of AutoDock
1.5.6, these compounds were docked into the active site cavity of human sex hormone-binding globulin (hSHBG) to evaluate
their binding affinity. Docking results showed that the binding affinities of BaA and its monohydroxy derivatives lie in the
comparable range (−8.5 kcal/mol to −9.04 kcal/mol) with dihydrotestosterone (DHT) (−10.94 kcal/mol), a known ligand of
hSHBG. The combined results from both the computational models emphasized that BaA and its metabolites can structurally
mimic the binding pattern of DHT, a known inhibitor to hSHBG.
Keywords
Benzo(a)anthracene Human sex hormone-binding globulin Density functional theory calculations Docking.References
No references available for this article.
Citation
Nikita Tiwari, Anil Mishra*. Computational Models: A Sustainable Approach to Reveal the Inhibitory Potential of Benzo(a)anthracene and its Monohydroxy Derivatives Against Human Sex Hormone-Binding Globulin. Indian J. Adv. Chem. Sci. 2022; 9(4):250-254.