Evaluation of Molecular Structure and Properties of Acetylacetone Thiosemicarbazone Using DFT Approach: Exploring Quantum Chemistry for Futuristic Applications
Karnica Srivastava, Tahmeena Khan
DOI: 10.22607/IJACS.2021.904003
Volume 9, Issue 4 | Pages: 261-267
Abstract
Thiosemicarbazone ligands are important Schiff bases and their utility has been studied since a long time. This paper presents
the evaluation of molecular structure and properties of 2-(4-oxopentan-2-ylidene) hydrazinecarbothioamide (acetylacetone
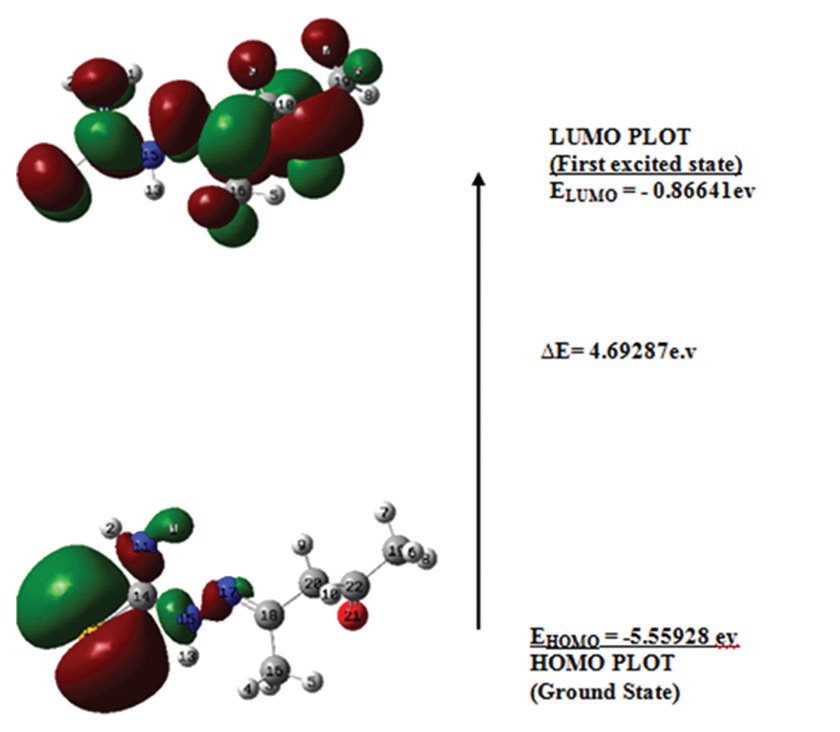
thiosemicarbazone) ligand using the density functional theory (DFT) calculations. B3LYP data set was used for the DFT
based evaluation. Primary hyperconjugative interactions were the donation of electron pair from a lone pair orbital LP(1)
N15→σ*S12-C14, LP(1)N15→π*N17-C18, and LP(2)O21→σ*C20-C22 leading to the stabilization of the ligand molecule with the
energy of 63.91, 28.16, and 22.74 kcal/mol, respectively. The molecular electrostatic potential (MEP) analysis showed that
hydrogen atoms act as nucleophilic centers and oxygen (O21) and sulfur (S12) atoms act as the electrophilic centers of ligand,
respectively. The non-linear optical (NLO) evaluation showed that the βo value calculated for ligand is 0.436 × 10−30 e.s.u.
which was better than the βo value of urea, that is, 0.1947 × 10−30 e.s.u. which is a standard NLO material. Therefore, the ligand
molecule might be used as NLO material in future.
Keywords
Thiosemicarbazone Schiff base Density function theory Non-linear optical and MEP surface analyses.References
No references available for this article.
Citation
Karnica Srivastava, Tahmeena Khan. Evaluation of Molecular Structure and Properties of Acetylacetone Thiosemicarbazone Using DFT Approach: Exploring Quantum Chemistry for Futuristic Applications. Indian J. Adv. Chem. Sci. 2022; 9(4):261-267.