Second Derivative Spectrophotometric Determination of Zinc(II) Using 2-Acetyl pyridine Thiosemicarbazone/Semicarbazone in Biological Samples
S. Vidyasagar Babu, K. S. V. Krishna Rao*
DOI: 10.22607/IJACS.2020.802007
Volume 8, Issue 2 | Pages: 72-77
Abstract
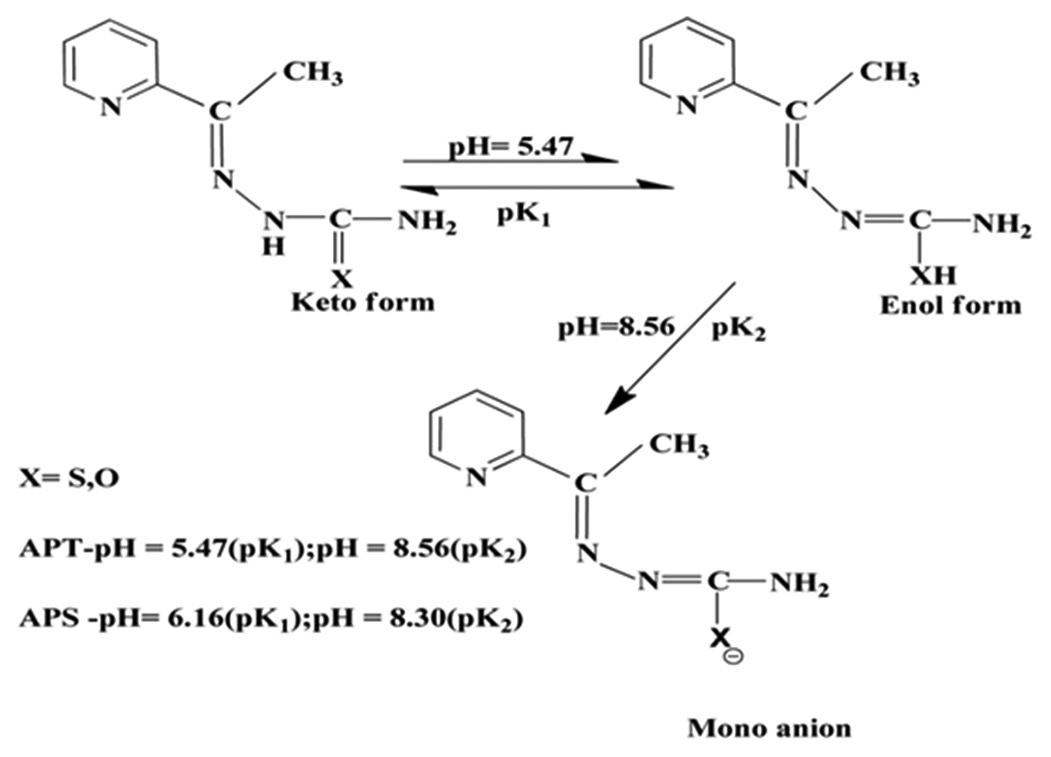
2-Acetylpyridine thiosemicarbazone (APT) and 2-acetylpyridine semicarbazone (APS) have been used for the spectrophotometric
determination of zinc(II) in aqueous medium. APT and APS react with zinc(II) in acidic medium. The color reactions between
reagents with zinc(II) are instantaneous and the absorbance of complexes remains constant for over 24 h. The maximum
absorbance (lmax), composition (M: L), molar absorptivity, and Sandells sensitivity of the Zn-APT and Zn-APS complexes,
respectively, are 360, 355 nm, 1:2, 4.06×104, 8.12×103 L/mol/cm, and 0.0160, 0.080 μgcm−2 of Zn(II), respectively. The Zn-
APT and Zn-APS systems obey Beer’s law for 0.105–1.046, and 0.523–5.231 μg/ml of Zn(II), respectively. Large number of
cations, anions, and complexing agents (e.g. triethanolamine, and thiourea) does not interfere in APT method. The method is
successfully applied for the determination of zinc in biological samples.
Keywords
Spectrophotometry Zinc determination 2-Acetylpyridine thiosemicarbazone 2-Acetylpyridine semicarbazone Biological samples.References
No references available for this article.
Citation
S. Vidyasagar Babu, K. S. V. Krishna Rao*. Second Derivative Spectrophotometric Determination of Zinc(II) Using 2-Acetyl pyridine Thiosemicarbazone/Semicarbazone in Biological Samples. Indian J. Adv. Chem. Sci. 2020; 8(2):72-77 .