Biosorptive Removal of Arsenate by Ficus benghalensis Stem Powder from Aqueous Medium
Ashish Kumar*, J. P. Pandey, Satish Kumar
DOI: 10.22607/IJACS.2020.804008
Volume 8, Issue 4 | Pages: 206-215
Abstract
The adsorption characteristics of As(V) on stem powder of Ficus benghalensis, a low-cost natural biosorbent, were studied
through batch mode of experimental investigation, resulting into reasonably remarkable As(V) removal efficiency over a wide
range of pH (3.0–11.0). The kinetics studies evidenced that the removal of As(V) by above-mentioned biosorbent which is favored
by pseudo-first-order kinetics. The equilibrium data of the adsorption were modeled using Langmuir, Freundlich, Temkin, and
DR isotherm. The adsorption isotherm was well described by Freundlich isotherm model, with maximum correlation coefficient
(R2 = 0.999), adsorption intensity (n = 1.920), and adsorption capacity (KF = 1.303). Thermodynamic parameters such as the
positive value of ∆H°, ∆S°, and negative results of ∆G° indicated that adsorption process is endothermic, increased randomness,
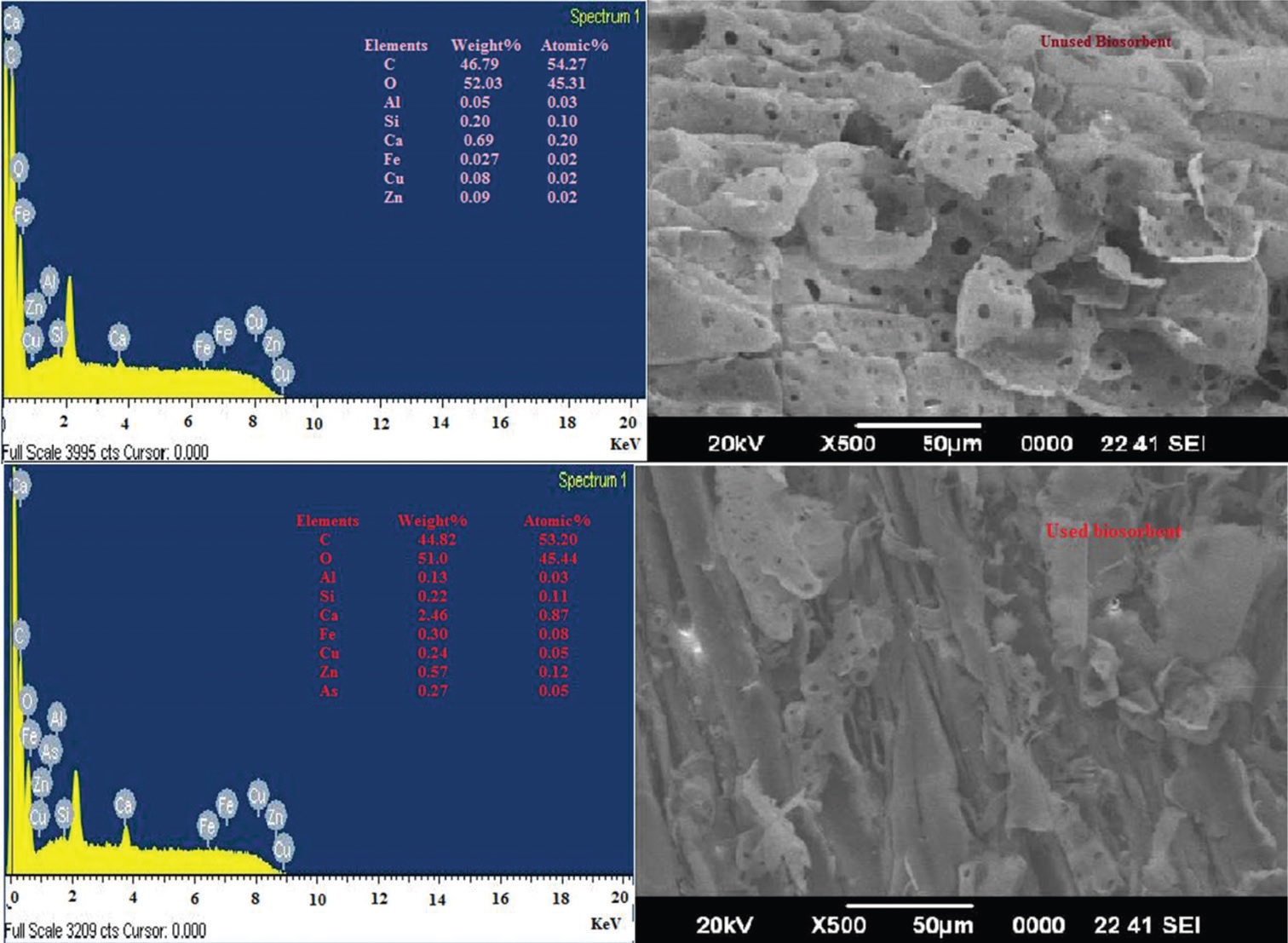
and spontaneous in nature, respectively. The presence of interfering ions such as anion F-, Cl-, NO3-, and SO42- significantly
reduces the As(V) removal efficiency of the biosorbent while cations such as Fe(III), Cu(II), Ni(II), Zn(II), Pb(II), Ca2+, and
K+ increases the adsorption efficiency. On the other hand, desorption studies revealed that As(V) can be easily desorbed by
(0.1 0.01 M) HCl and HNO3 and such biosorbent can be reused.
Keywords
Arsenic Biosorbent Adsorption isotherm Stem powder of Ficus benghalensis Desorption.References
No references available for this article.
Citation
Ashish Kumar*, J. P. Pandey, Satish Kumar. Biosorptive Removal of Arsenate by Ficus benghalensis Stem Powder from Aqueous Medium. Indian J. Adv. Chem. Sci. 2021; 8(4):206-215.