Synthesis, Crystal Structure, and Characterization of Manganese Metal-organic Framework with Improved Electro Catalytic Activity for Supercapacitor Application
G. Krishnamurthy*, B. M. Omkaramurthy
DOI: 10.22607/IJACS.2019.701002
Volume 7, Issue 1 | Pages: 12-19
Abstract
A manganese metal-organic framework (Mn-MOF) has been synthesized using a low-temperature solvothermal procedure in
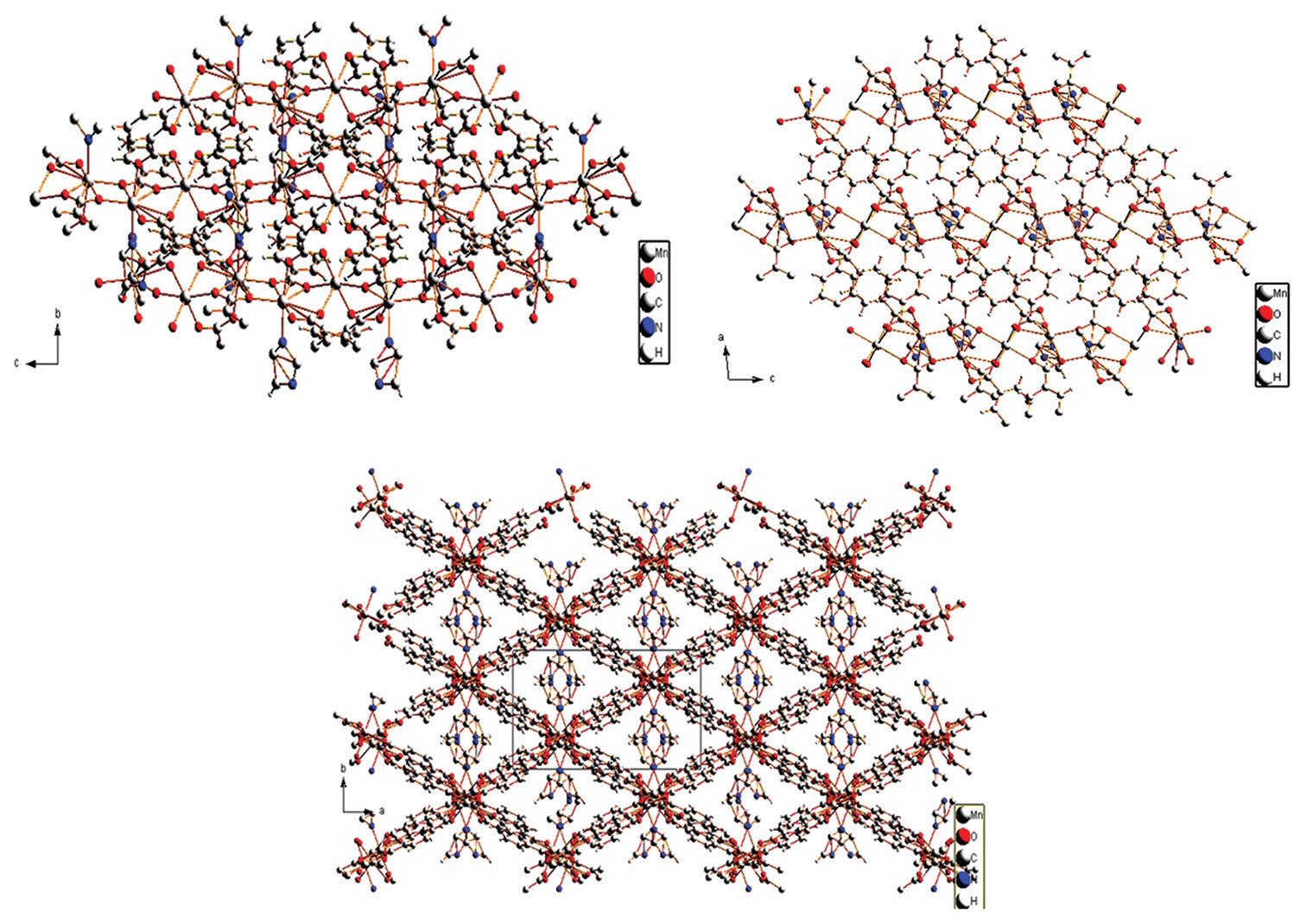
the presence of terephthalic acid and 1-methylimidazole in dimethylformamide. The crystal structure studies were made and
found that the molecule has a three dimensional framework in which the θ ranges from 5.942 to 50 with monoclinic I2/a space
group. The final full matrix least-square refinement over F2 is converged on R1 = 0.0558, wR2 = 0.1696 through goodness-of-
fit = 1.095. The surface area of the Mn-MOF has been measured by Brunauer–Emmett–Teller method and the surface area
was found to be 1.2352 m2/g. The average pore diameter and pore volume were about 2.6 nm and 0.00569 cc/g, respectively.
The electrochemical studies were made by cyclic voltammetry, galvanostatic charge-discharge, and electrochemical impedance
spectroscopy techniques and found that the Mn-MOF has high charge storage capacity up to 4000 Fg-1, which reflects the
supercapacitor behaviour of the material. Also Mn-MOF material exhibits the energy density up to 500 (Whkg-1) and the
power density of about 0.125 (Wkg-1). These values imply that the Mn-MOF can be an efficient material for electrochemical
supercapacitor/other energy storage application.
Keywords
Manganese metal-organic framework Secondary building units Solvothermal synthesis Supercapacitor Cyclic voltammetry.References
No references available for this article.
Citation
G. Krishnamurthy*, B. M. Omkaramurthy. Synthesis, Crystal Structure, and Characterization of Manganese Metal-organic Framework with Improved Electro Catalytic Activity for Supercapacitor Application. Indian J. Adv. Chem. Sci. 2019; 7(1):12-19.