Validated Spectrophotometric Method for the Determination of Lumefantrine in Pharmaceutical Formulation by Charge-Transfer Complexation with Picric Acid
Ukpe Ajima*, Johnson O. Onah, Bilma W. Obidah, Paul N. Olotu
DOI: 10.22607/IJACS.2019.702002
Volume 7, Issue 2 | Pages: 46-51
Abstract
Lumefantrine is an aryl amino alcohol derivative widely used in combination with artemether for the therapy of malaria. A simple
and accurate spectrophotometric method has been developed and validated for the quantitative determination of lumefantrine
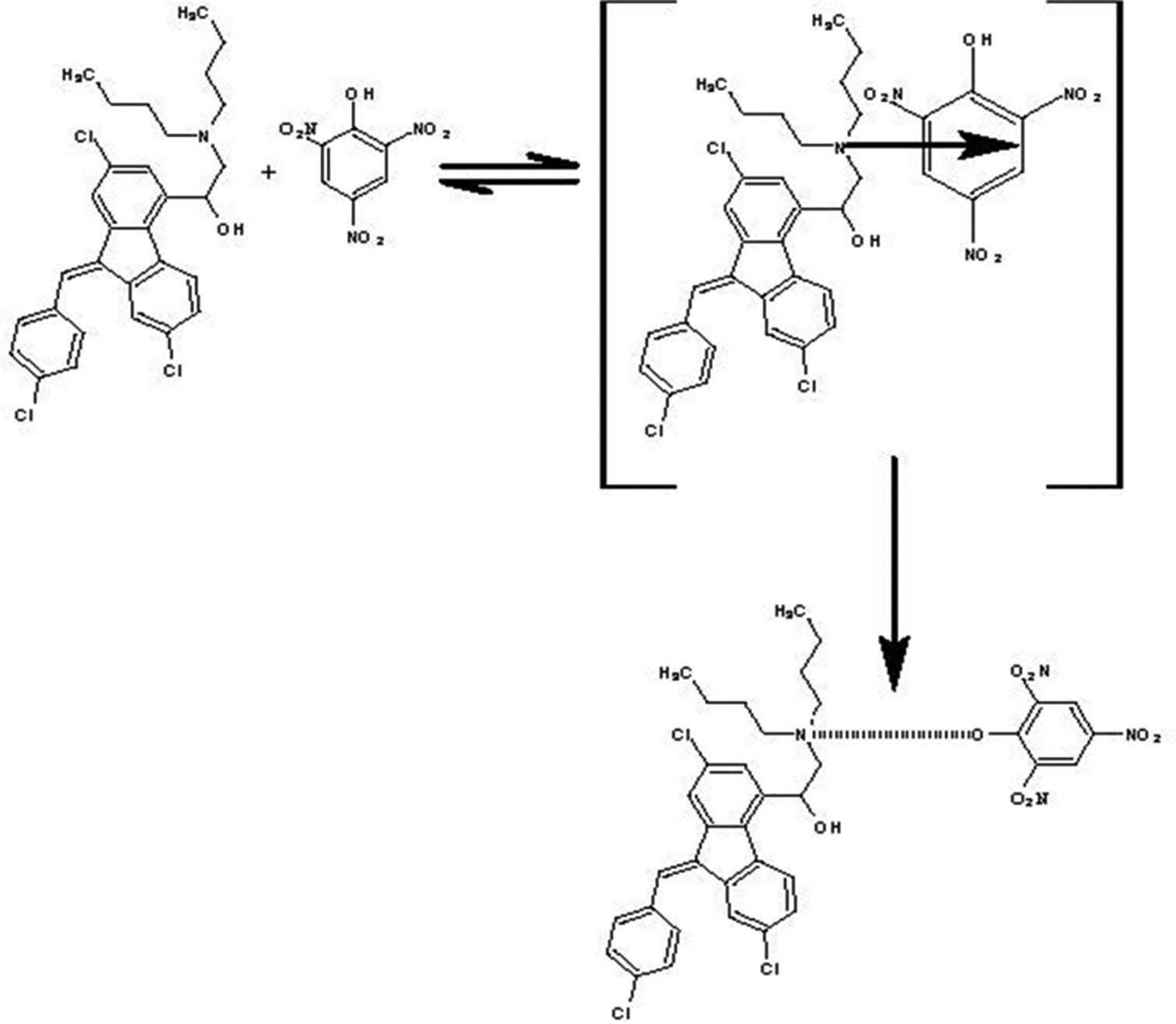
in finished pharmaceutical products. The proposed method is based on the charge-transfer (CT) reaction of lumefantrine (as
n-electron donor) with picric acid (2, 4, 6-trinitrophenol) as π-acceptor to give a highly colored CT complex. The different
variables affecting the reaction such as reagent concentration, dilution solvent, and reaction time were carefully optimized to
achieve the highest sensitivity. The lumefantrine-picric acid CT complex was quantified spectrophotometrically at 420 nm, and
Beer’s law was obeyed over the concentration range of 20–100 μg/mL with correlation coefficient (r2) of 0.9981. The limit of
detection and quantification were found to be 0.884 μg/mL and 1.507 μg/mL, respectively, for the method. The proposed method
was also found to give comparable results with the official method as no statistically significant differences were found in the
percentage drug content for five brands assayed by both methods. The developed method can, therefore, be successfully applied
to analyze lumefantrine content in both raw materials and finished pharmaceutical products.
Keywords
Lumefantrine Spectrophotometry Picric acid Charge transfer complex Method development.References
No references available for this article.
Citation
Ukpe Ajima*, Johnson O. Onah, Bilma W. Obidah, Paul N. Olotu. Validated Spectrophotometric Method for the Determination of Lumefantrine in Pharmaceutical Formulation by Charge-Transfer Complexation with Picric Acid. Indian J. Adv. Chem. Sci. 2019; 7(2):46-51.