In Silico Investigation of Charge Transfer and Non-covalent Interactions between Nitro-polycyclic Aromatic Hydrocarbons and DNA/RNA Bases and Base Pairs using Density Functional Theory
Mohmedyasin F. Mansuri, Shravan B. Rathod*
DOI: 10.22607/IJACS.2019.703003
Volume 7, Issue 3 | Pages: 76-87
Abstract
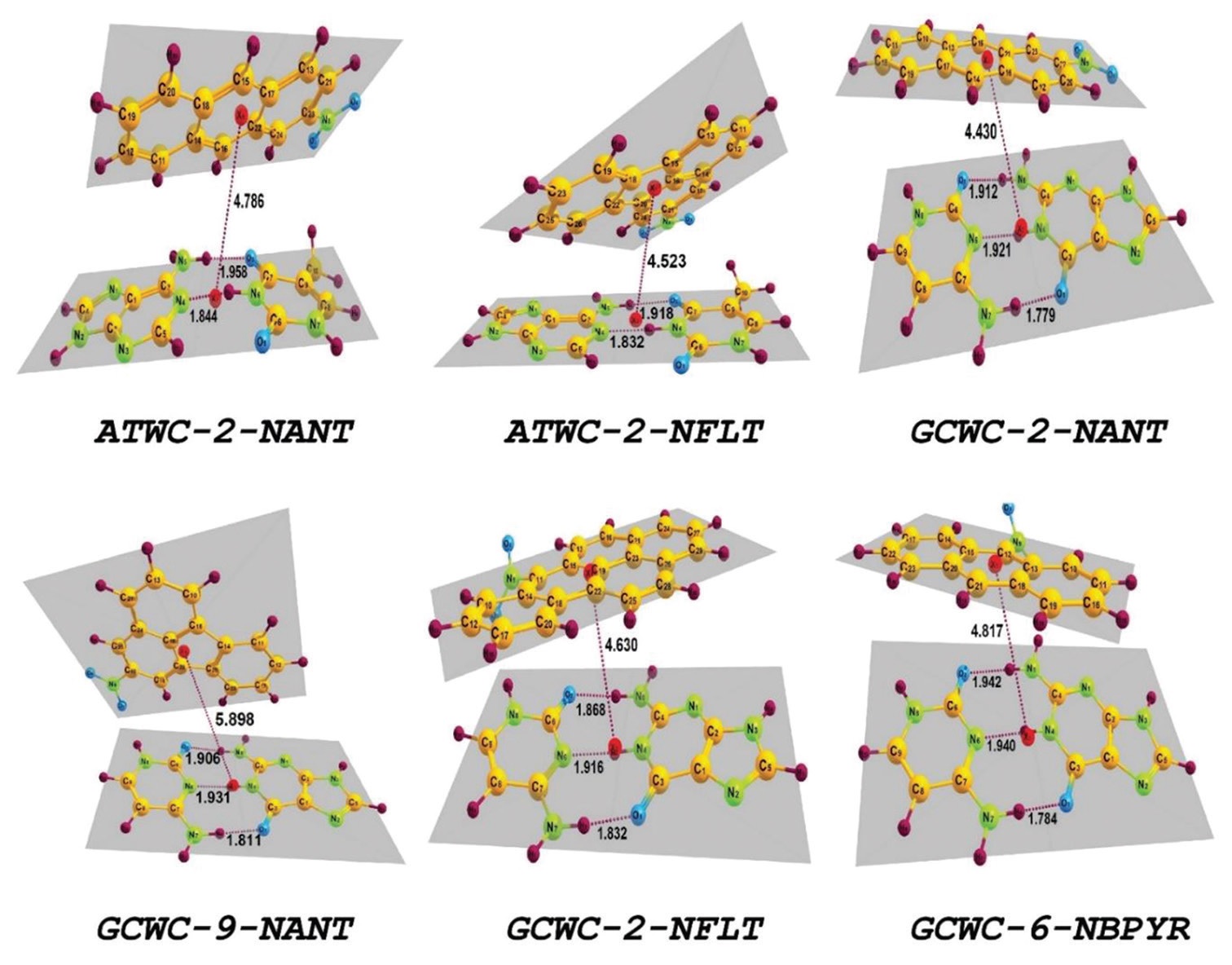
The electrophilicity-based charge transfer (ECT) between nine nitro-polycyclic aromatic hydrocarbons (NPAHs) and seven
DNA/RNA bases and base pairs was investigated and compared using ΔN and ECT methods at density functional theory level
theory. The ground-state geometries of molecules were optimized at density hybrid functional B3LYP and Pople basis set 6-31
G*. ΔN and ECT methods predict different results for charge transfer, and ECT method has a considerably higher amount of
charge transfer between two systems compared to ΔN method. Binding energies of NPAHs with Guanine-cytosine watson-crick
(GCWC) and Adenine-thymine watson-crick (ATWC) base pairs were calculated and then corrected using B3LYP-gCP-D3/6-
31G* scheme. The topological analysis of electron charge density was performed to investigate the non-covalent interactions
between NPAHs and DNA/RNA bases and base pairs using atoms in molecules (AIM) approach.
Keywords
Particulate matter2.5 Carcinogenic Toxins Electrophilicity index Binding energy.References
No references available for this article.
Citation
Mohmedyasin F. Mansuri, Shravan B. Rathod*. In Silico Investigation of Charge Transfer and Non-covalent Interactions between Nitro-polycyclic Aromatic Hydrocarbons and DNA/RNA Bases and Base Pairs using Density Functional Theory. Indian J. Adv. Chem. Sci. 2019; 7(3):76-87.