Spectrophotometric Method for the Estimation of Pantoprazole Sodium Using Reagents – 2,3-Dichloro-5,6-dicyano-1,4-benzoquinone, Chloranilic Acid, and 1-Chloro-2,4-dinitrobenzene with Charge – Transfer Complex Reaction
N. Subrahmanyeswara Rao, S. Arul Antony*, T. Thirupathi Rao, S. Siva Kumar
DOI: 10.22607/IJACS.2019.704002
Volume 7, Issue 4 | Pages: 111-115
Abstract
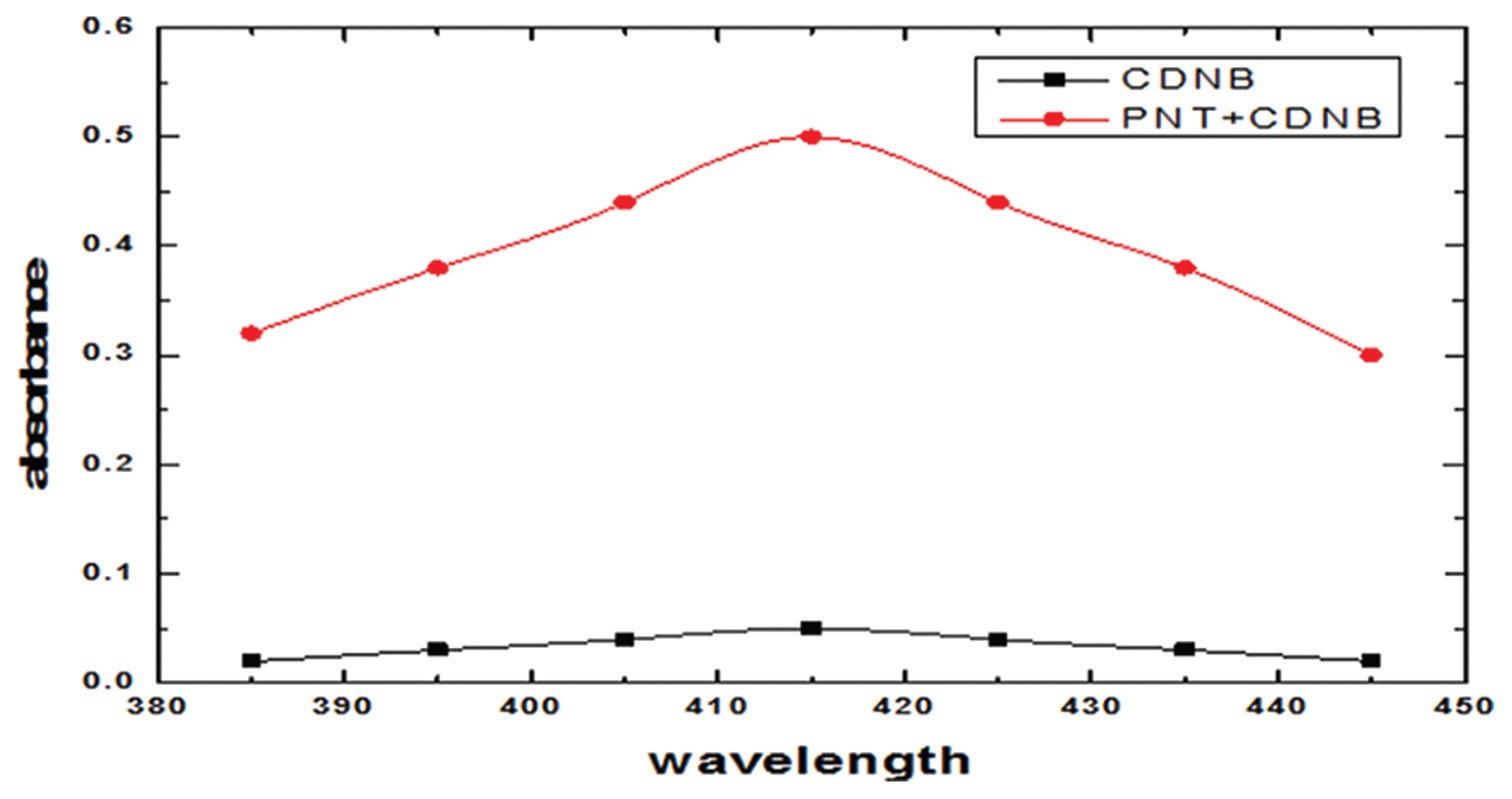
Three simple, sensitive, and rapid spectrophotometric methods were developed for the estimation of pantoprazole sodium (PNT)
in pure and its commercial dosage forms. These methods were based on the formation of charge–transfer reaction between
the drug, an n-electron donor and π-acceptors, 2, 3-dichloro-5, 6-dicyano-p-benzoquinone, chloranilic acid, and 1-chloro-2,
4-dinitrobenzene. The absorbance of the formed charge–transfer complexes was measured and utilized for the determination
of PNT in its pure and commercial dosage forms. The developed methods were evaluated in terms of standard deviation,
relative standard deviation, correlation coefficient, limit of detection, and limit of quantitation. Molar absorptivity and Sandell’s
sensitivity were calculated at the optimum experimental conditions. The validity of the proposed methods was ascertained by
recovery studies which indicated that the present methods can be successfully applied for the determination of PNT in pure and
commercial dosage forms.
Keywords
Spectrophotometric methods Pantoprazole sodium Charge–transfer reaction 2 3-Dichloro-5 6-dicyano-p- benzoquinone Chloranilic acid 1-Chloro-2 4-dinitrobenzene.References
No references available for this article.
Citation
N. Subrahmanyeswara Rao, S. Arul Antony*, T. Thirupathi Rao, S. Siva Kumar . Spectrophotometric Method for the Estimation of Pantoprazole Sodium Using Reagents – 2,3-Dichloro-5,6-dicyano-1,4-benzoquinone, Chloranilic Acid, and 1-Chloro-2,4-dinitrobenzene with Charge – Transfer Complex Reaction. Indian J. Adv. Chem. Sci. 2020; 7(4):111-115.