Gout Pain Caused by Uric Acid Molecule and Its Solution Caused by Citric Acid Molecule: A Physicochemical Investigation
Mahendra Nath Roy*, Subhankar Choudhury, Deepak Ekka, Raja Ghosh
DOI: 10.22607/IJACS.2018.602001
Volume 6, Issue 2 | Pages: 59-70
Abstract
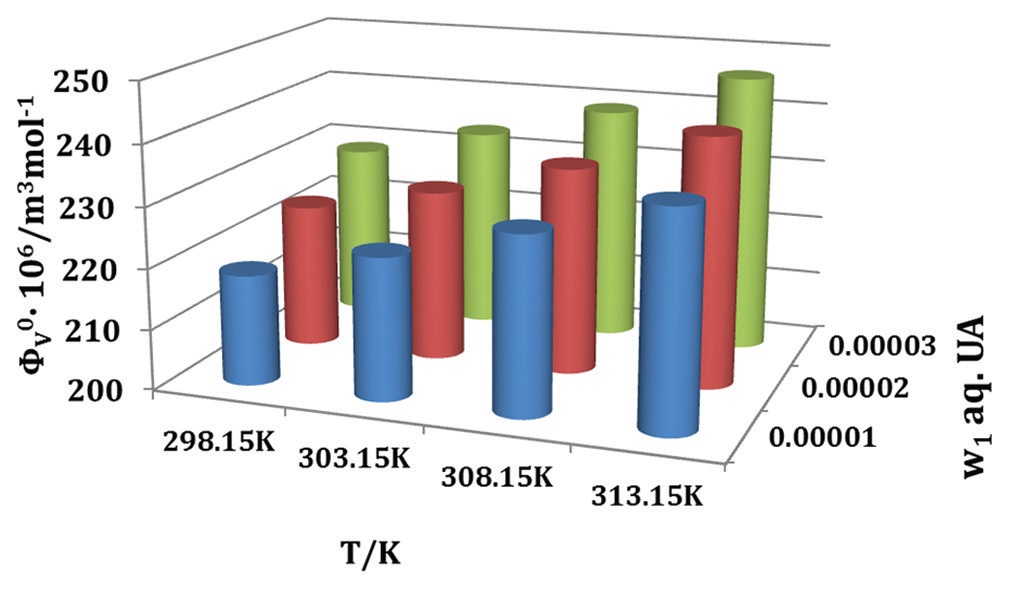
The effect of relief from gout pain due to the restriction of precipitation of uric acid (UA) by citric acid (CA) has been studied
through physicochemical study. Here, we have carried out the density (ρ) and viscosity (η) measurements of CA in w1=0.00001,
0.00002, and 0.00003 mass fraction of aqueous UA binary mixtures at T=298.15K, 303.15K, 308.15K, and 313.15K and at
pressure 1.013 bar. These measurements have been performed to ternary mixture (CA + UA + water) to derive some important
parameters, namely, limiting apparent molar volume (φV0), viscosity B-coefficients from extended Masson equation, and
Jones–Dole equation, respectively. The refractive index (nD) has been done on the same ternary mixtures at T=298.15K.
Lorentz–Lorenz equation has used to evaluate molar refractive index (RM) and limiting molar index (RM0). These parameters
have been interpreted in terms of interactions of solute itself and with solvent.
Keywords
Apparent molar volume Citric acid Molar refraction Solute-solvent interactions Uric acid Viscosity B-coefficient.References
No references available for this article.
Citation
Mahendra Nath Roy*, Subhankar Choudhury, Deepak Ekka, Raja Ghosh. Gout Pain Caused by Uric Acid Molecule and Its Solution Caused by Citric Acid Molecule: A Physicochemical Investigation. Indian J. Adv. Chem. Sci. 2018; 6(2):59-70 .