Adsorptive Removal of Copper and Nickel Ions from Aqueous using Chitosan-g-Poly(Acrylic Acid)/Polyaniline Membrane
T. J. Sudha Vani, K. S. V. Krishna Rao*
DOI: 10.22607/IJACS.2018.602003
Volume 6, Issue 2 | Pages: 77-82
Abstract
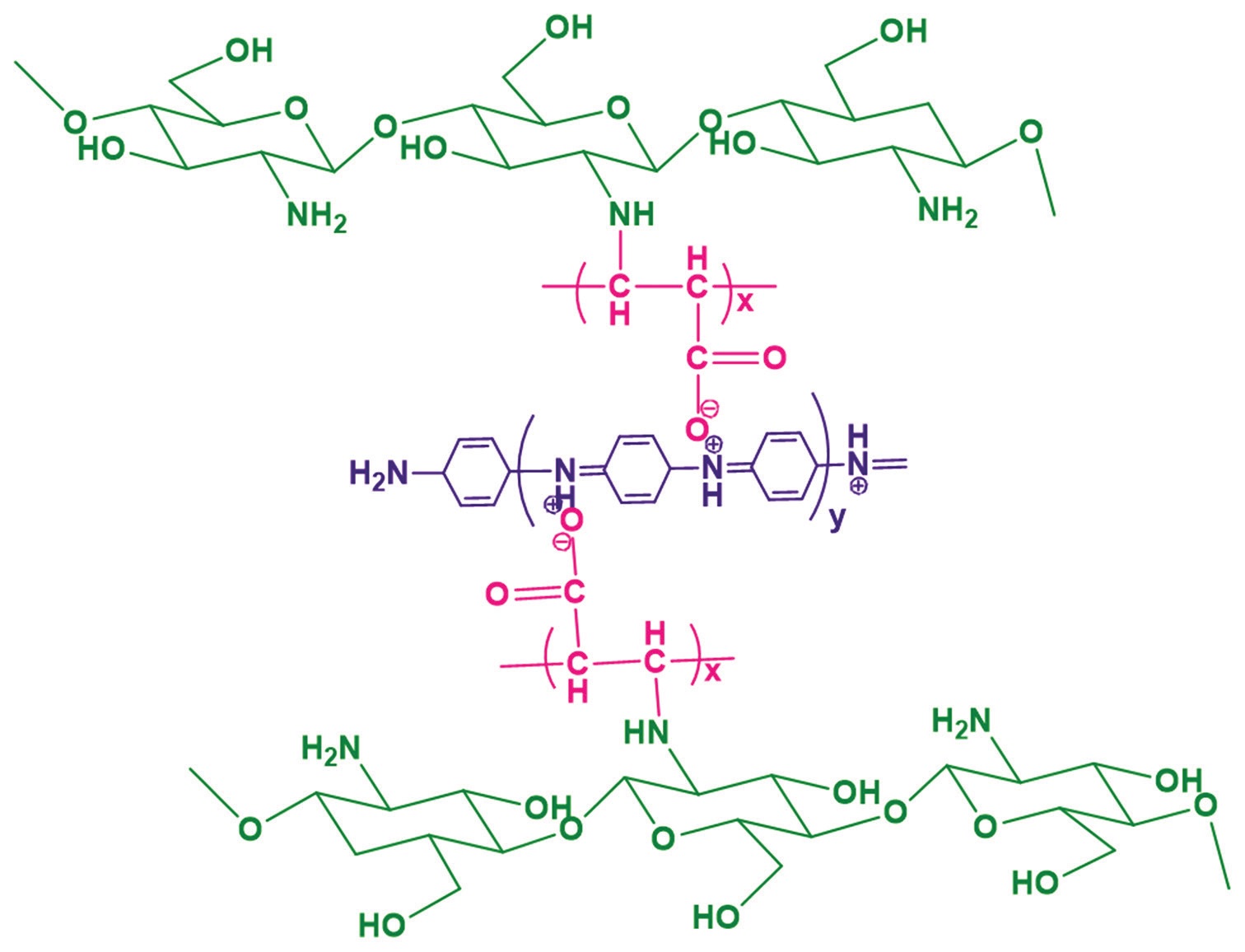
The present work reports the development of new chitosan-based membrane by grafting with poly(acrylic acid) and polyaniline
by free radical polymerization using ammonium persulfate. Grafting reaction of chitosan-g-poly(acrylic acid)-co-polyaniline
(CPANI) was confirmed by Fourier-transform infrared spectroscopy, surface morphology of membranes was characterized by
scanning electron microscopy. The influence of various experimental conditions such as pH, time, and initial feed concentrations
on the uptake of Cu2+ and Ni2+ by CPANI membrane was tested. The optimum pH was found to be 5.0. It was seen that the
adsorption equilibrium data could be fitted to the Langmuir isotherm. Desorption studies were performed in acid media and
ethylenediaminetetraacetic acid, to examine whether the CPANI membrane can be recycled for the metal ion removal. The
results showed that with CPANI membrane, the maximum metal ion uptake achieved is 1.01 mM g−1 for Cu2+ and 2.41 mM g−1
for Ni2+,respectively. Metal ion sorption studies showed that the membrane can be used for the removal of hazardous metal ions
from aqueous solutions. The copper and nickel uptake achieved suggests the potential use of the membrane to extract divalent
toxic metals from industrial aqueous streams.
Keywords
Chitosan Copper Membrane Nickel Poly(Acrylic acid) Polyaniline.References
No references available for this article.
Citation
T. J. Sudha Vani, K. S. V. Krishna Rao*. Adsorptive Removal of Copper and Nickel Ions from Aqueous using Chitosan-g-Poly(Acrylic Acid)/Polyaniline Membrane. Indian J. Adv. Chem. Sci. 2018; 6(2):77-82 .