Removal of Fluoride from Aqueous Solution by Adsorption on Perchloric Acid Cross-linked Calcium Alginate
Vijaya Yarramuthi*, Jaya Malathi Jagadeeson, Karuna Devi Rachapudi, Raja Kumar Challagundla
DOI: 10.22607/IJACS.2018.602005
Volume 6, Issue 2 | Pages: 88-95
Abstract
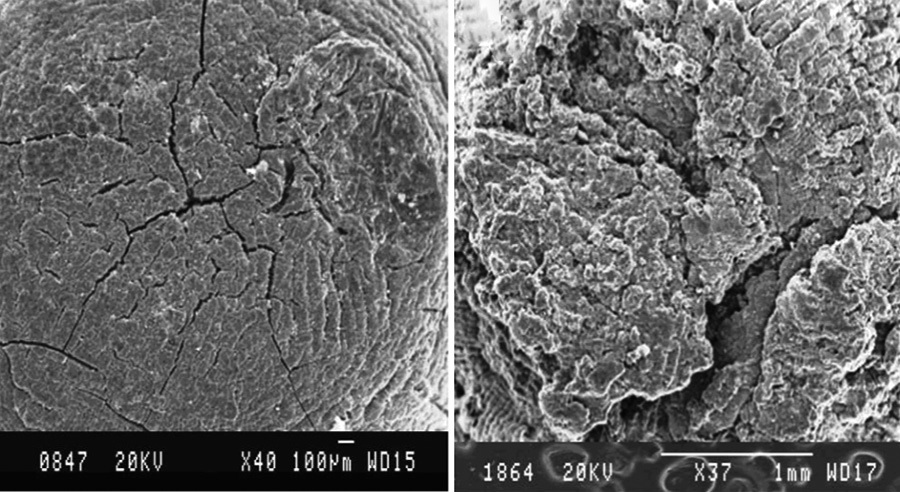
A novel biosorbent was developed by the crosslinking of an anionic biopolymer, calcium alginate (CA), with perchloric acid
(PCA). The PCA cross-linked was characterized by Fourier-transform infrared, scanning electron microscopy (SEM), trans-
mission electron microscopy, X-ray diffraction, and surface area analysis. Experimental parameters affecting the adsorption
process such as pH, agitation time, concentration of adsorbate, and amount of adsorbent were studied. Langmuir and Freundlich
isotherms were used to fit the experimental data. The best interpretation for the equilibrium data was given by the Langmuir
isotherm. The maximum adsorption capacity for fluoride was 44 mg/g based on the Langmuir equation at a temperature of 28°C,
a solution pH of 3.0, a adsorbent dosage of 0.7 g/100 mL, and a contact time of 120 min. The experimental data were analyzed
using three sorption kinetic models, namely, pseudo-first-order, pseudo-second-order, and intraparticle diffusion model. Results
show that the pseudo-second-order equation provides the best correlation for the adsorption process. The findings of the present
study indicate that PCA can be successfully used for the removal of fluoride ions from aqueous solutions.
Keywords
Perchloric acid Defluoridation Adsorption Isotherms Kinetics.References
No references available for this article.
Citation
Vijaya Yarramuthi*, Jaya Malathi Jagadeeson, Karuna Devi Rachapudi, Raja Kumar Challagundla. Removal of Fluoride from Aqueous Solution by Adsorption on Perchloric Acid Cross-linked Calcium Alginate. Indian J. Adv. Chem. Sci. 2018; 6(2): 88-95 .