Exploring the Molecular Interactions of Nicotinic Acid Prevailing in Salicylic Acid + H2O, Anthranilic Acid + H2O and o-Nitrobenzoic Acid + H2O Mixed System at Different Temperatures
Kalipada Sarkar, Mahendra Nath Roy*
DOI: 10.22607/IJACS.2018.603001
Volume 6, Issue 3 | Pages: 96-106
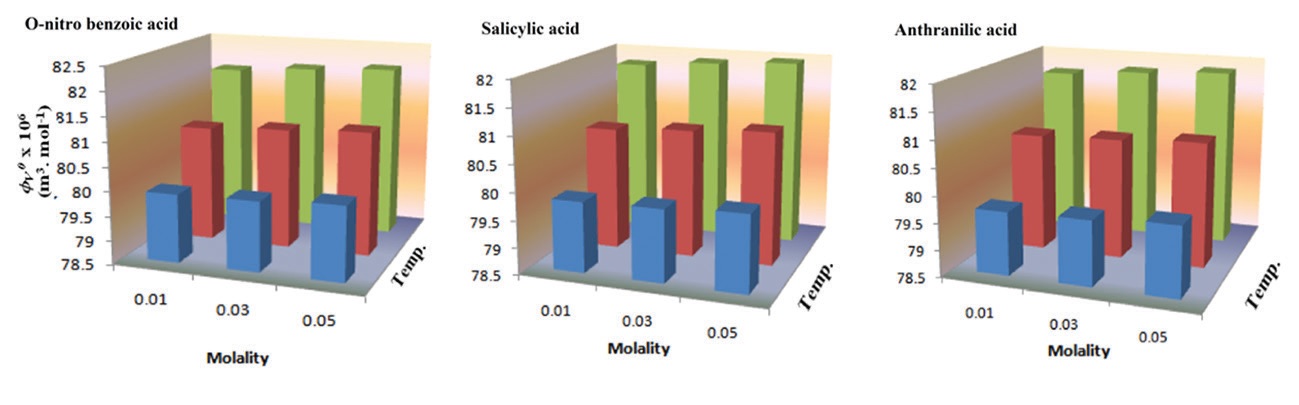
Abstract
Molecular interactions of some o-substituted benzoic acids, namely, salicylic acid, anthranilic acid, and o-nitrobenzoic acid with
nicotinic acid are examined by physicochemical properties as density (ρ) and viscosity (η) at 298 K, 203 K, and 208 K. The
limiting apparent molar volume (φV0) and experimental slopes (SV*) determined from the Masson equation have been interpreted
in terms of solute-solvent and solute-solute interactions, respectively. The limiting apparent molar volume of transfer (∆φV0) has
been determined to examine the ionic-ionic, hydrophilic-hydrophilic, and hydrophobic-hydrophobic interaction taking place in
the solution. The viscosity data were examined using the Jones–Dole equation and the parameters viscosity A and B coefficients
have also been interpreted in terms of solute-solute and solute-solvent interactions, respectively. Molar free energy of activation of
viscous flow of the solvent, ∆μ10 and solute, ∆μ2° has been deduced to understand the stability of ground state over transition state.
Keywords
Nicotinic acid Limiting apparent molar volume Solute-solvent interaction Viscosity A and B coefficients Hydrophobic- hydrophobic interactionReferences
No references available for this article.
Citation
Kalipada Sarkar, Mahendra Nath Roy*. Exploring the Molecular Interactions of Nicotinic Acid Prevailing in Salicylic Acid + H2O, Anthranilic Acid + H2O and o-Nitrobenzoic Acid + H2O Mixed System at Different Temperatures. Indian J. Adv. Chem. Sci. 2018; 6(3):96-106.