Novel Validated Stability-indicating Ultra-performance Liquid Chromatography Method for the Determination of Roflumilast and its Degradation Products in Active Pharmaceutical Ingredient and in Pharmaceutical Dosage Forms
Varaprasad Jagadabi, P. Venkata Nagendra Kumar*, Srinivasu Pamidi, L. A. Ramaprasad, R. Ramakrishna
DOI: 10.22607/IJACS.2018.603007
Volume 6, Issue 3 | Pages: 142-148
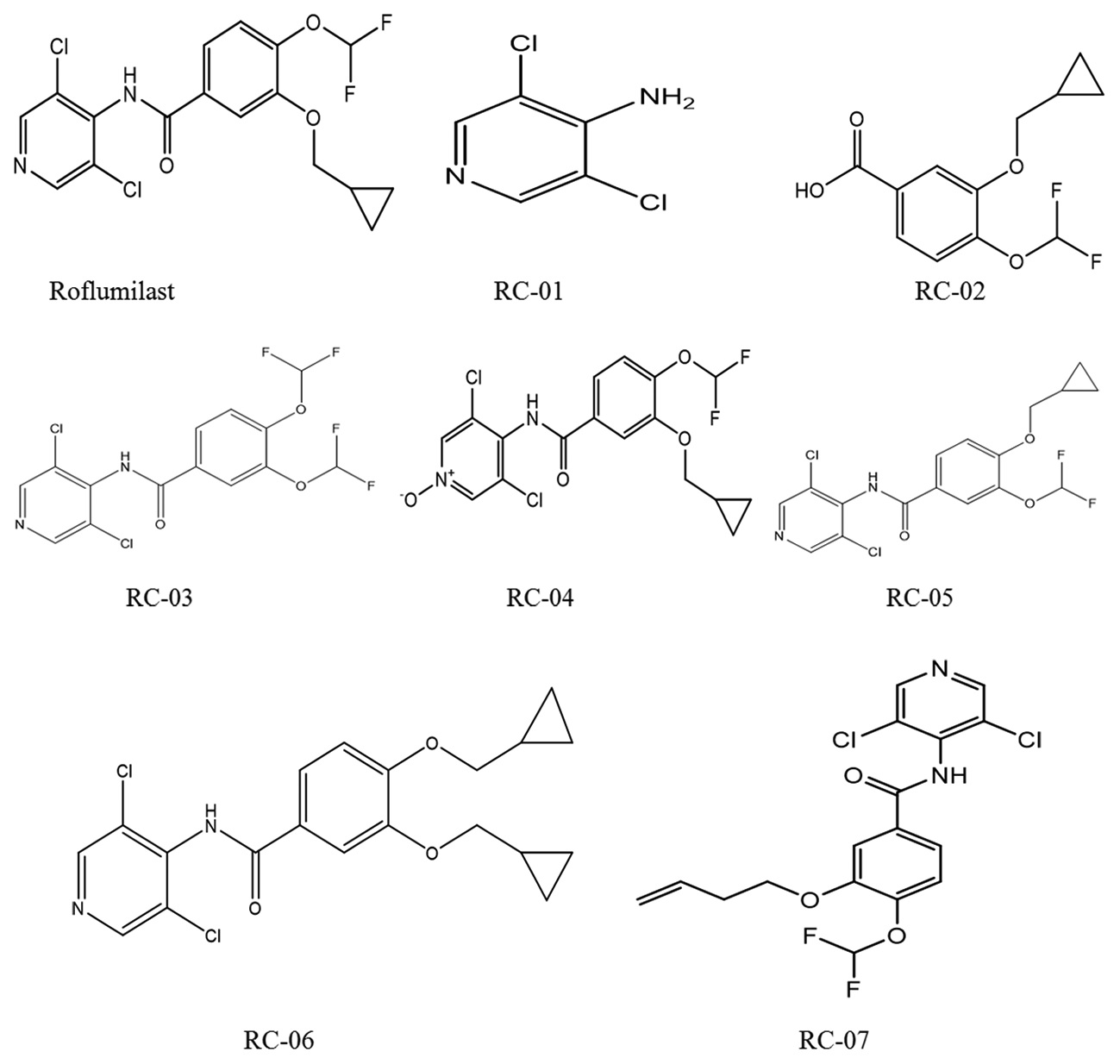
Abstract
A novel stability-indicating reverse-phase ultra-performance liquid chromatographic method was developed for quantitative
determination of roflumilast and its related impurities and degradation products. Chromatographic separation was achieved
using a waters acquity ultra-performance liquid chromatography BEH C18 100 mm × 2.1 mm, 1.7 μ column with mobile phase
containing a gradient mixture of mobile phase A and B at 60°C with a flow rate of 0.4 mL/min. The related compounds were
monitored at 220 nm. The run time was 15 min within which roflumilast and its seven related impurities were well resolved.
The developed method was validated as per ICH guidelines with respect to specificity, linearity, limit of detection, limit of
quantification, accuracy, precision, and robustness. The calibration curves obtained for the seven impurities were linear over the
range of 0.202–3.880 μg/mL. The relative standard deviations of intra- and inter-day experiments were <3.0%. The detection
limits ranged from 0.070 to 0.085 μg/mL depending on the impurity.
Keywords
Roflumilast Method validation Potential degradation products Impurities Stability indicating Ultra-performance liquid chromatography.References
No references available for this article.
Citation
Varaprasad Jagadabi, P. Venkata Nagendra Kumar*, Srinivasu Pamidi, L. A. Ramaprasad, R. Ramakrishna. Novel Validated Stability-indicating Ultra-performance Liquid Chromatography Method for the Determination of Roflumilast and its Degradation Products in Active Pharmaceutical Ingredient and in Pharmaceutical Dosage Forms . Indian J. Adv. Chem. Sci. 2018; 6(3):142-148.