Synthesis and Determination of Antioxidant Activity of Alizarin Derivatives
Chinnakurli Dwarkanath Shruthi, Vijeth Rajshekar Shetty, Gurukar Shivappa Suresh
DOI: 10.22607/IJACS.2018.604002
Volume 6, Issue 4 | Pages: 182-186
Abstract
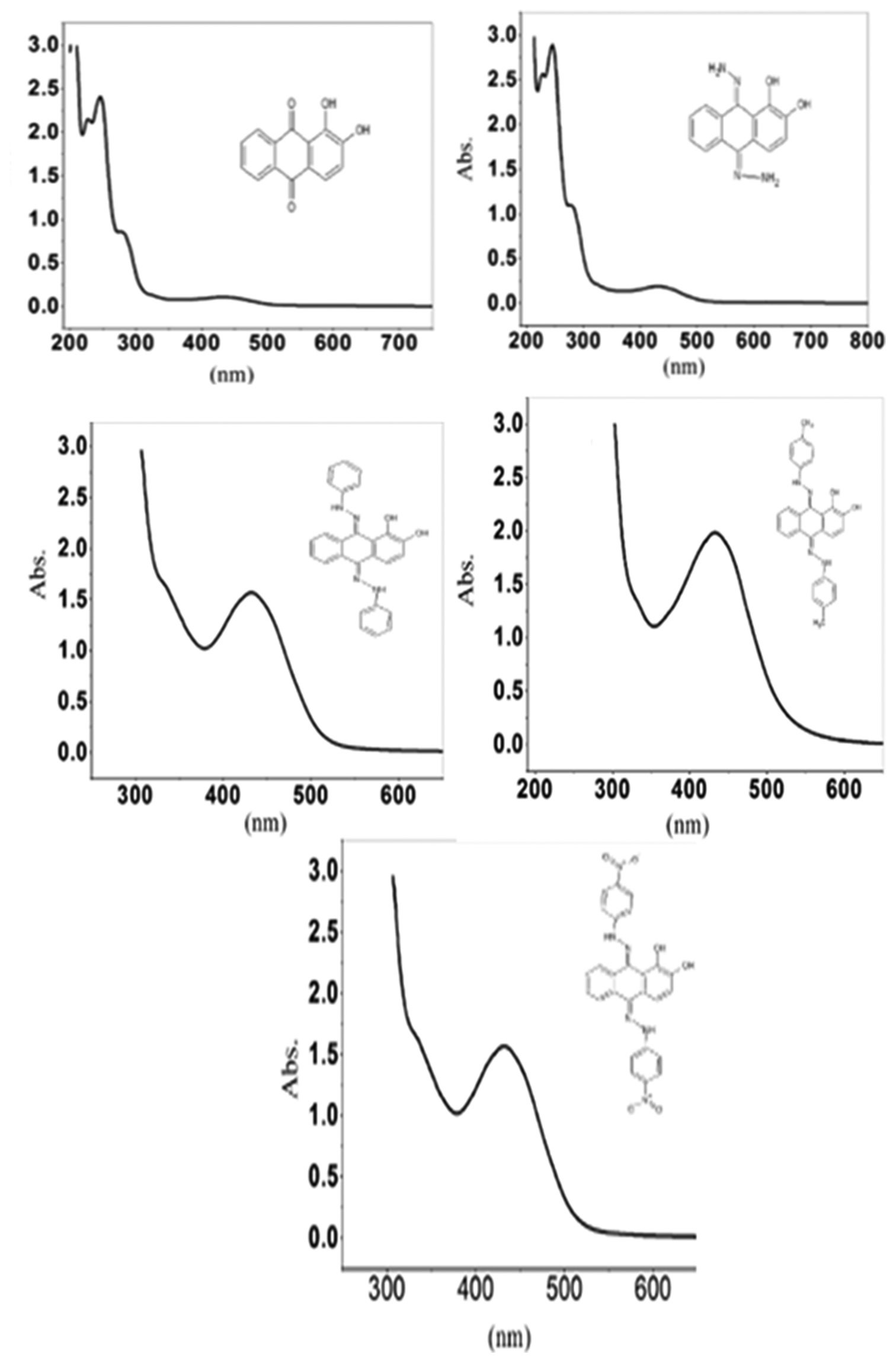
In the present work, we have synthesized nitro, phenyl, nitrophenyl, and methyl phenylhydrazone derivatives of alizarin and compared
their antioxidant activity. The antioxidant activities of each derivative were compared using electrochemical assay with glassy carbon
electrodes. The X-ray diffraction studies of compounds revealed that prominent peaks at 43 2q and 49 2q confirm the formation
alizarin, whereas the intensity varies accordingly with the functional groups attached to it. This was further confirmed by H1 nuclear
magnetic resonance. The antioxidant activities were interpreted by cyclic voltammetry that depends on the parameters such as anodic
peak current, anodic peak potential, and area swept by the curve. The maximum antioxidant activity was observed in phenylhydrazone
derivative of alizarin. The area of the peak and peak potential was observed at 108.017 μc and 347.498 mv, respectively. Hence, the
synthesis of the above derivatives of alizarin clearly explains the influence of functional groups on antioxidant activity.
Keywords
Antioxidant Activity Alizarin Phenylhydrazone.References
No references available for this article.
Citation
Chinnakurli Dwarkanath Shruthi, Vijeth Rajshekar Shetty, Gurukar Shivappa Suresh. Synthesis and Determination of Antioxidant Activity of Alizarin Derivatives. Indian J. Adv. Chem. Sci. 2019; 6(4):182-186.