Amberlite Infrared-120 Catalyzed Synthesis of 6-Aryl-5H-Quinazolino[4,3-b]Quinazolin8(6H)-one Derivatives as Anticancer Agents
Varimadugu Aruna, H. Sudhakar, Gangadhar Thalari*, Naveen Mulakayala
DOI: 10.22607/IJACS.2018.604003
Volume 6, Issue 4 | Pages: 187-192
Abstract
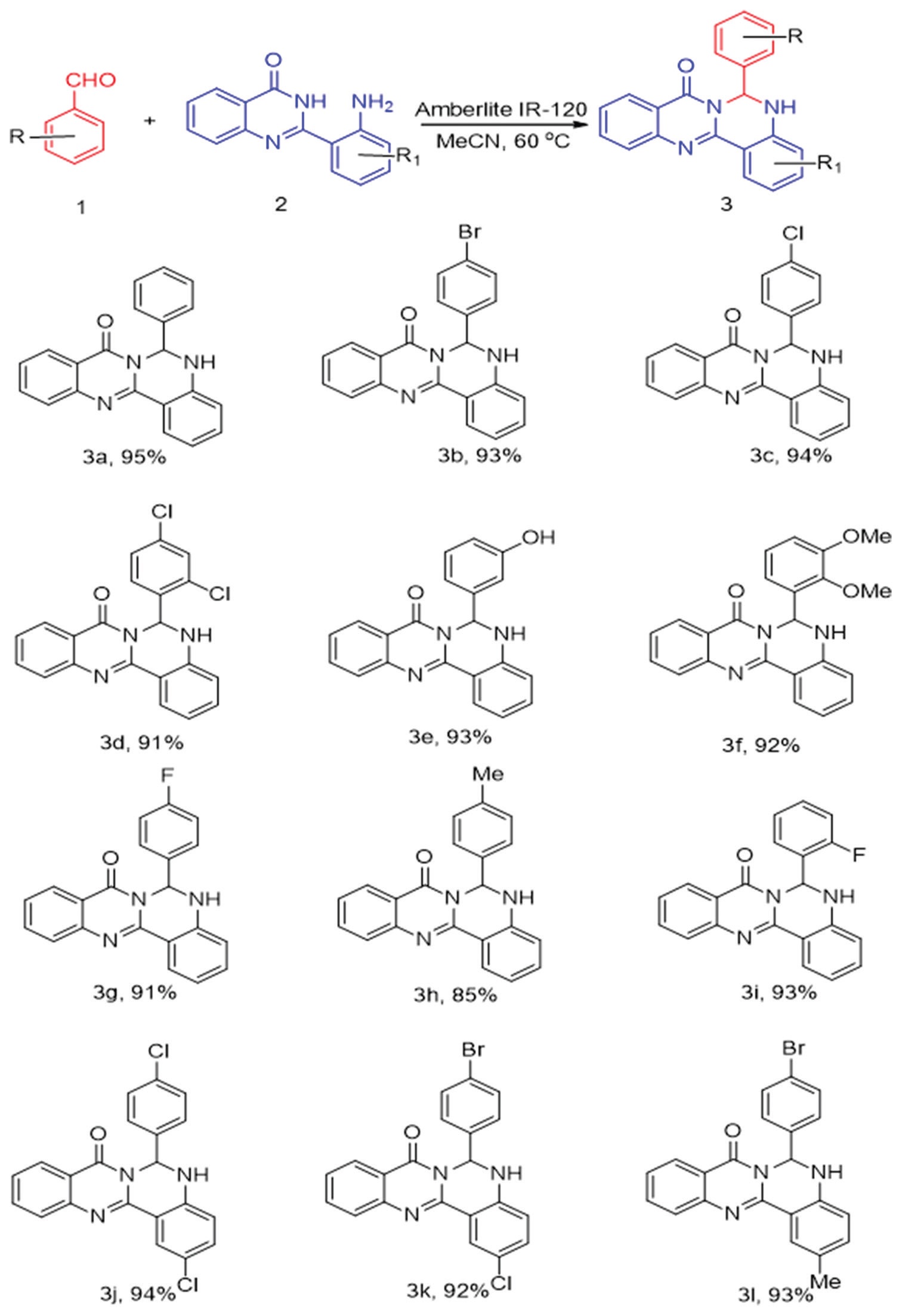
Amberlite infrared (IR)-120 catalyzed synthesis of 6-aryl-5H-quinazolino[4,3-b]quinazolin8(6H)-one derivatives was reported.
2-(2-aminophenyl)quinazolin-4(3H)-ones on reaction with aromatic aldehydes using Amberlite IR-120 resin yielded a variety
of 6-aryl-5H-quinazolino[4,3-b]quinazolin-8(6H)-one derivatives in good to excellent yields. This method has the advantages
of high yields, easy purification and having mild reaction condition. All the synthesized compounds were evaluated for their
anti-proliferative properties in vitro against cancer cell lines, and several compounds were found to be active.
Keywords
Amberlite IR-120 2-(2-Aminophenyl)quinazolin-4(3H)-ones Aromatic aldehydes Synthesis High yield.References
No references available for this article.
Citation
Varimadugu Aruna, H. Sudhakar, Gangadhar Thalari*, Naveen Mulakayala . Amberlite Infrared-120 Catalyzed Synthesis of 6-Aryl-5H-Quinazolino[4,3-b]Quinazolin8(6H)-one Derivatives as Anticancer Agents. Indian J. Adv. Chem. Sci. 2019; 6(4):187-192.