Removal of methylene blue from wastewater using Fe2O3 as an adsorbent
Ajoy Kanti Mondal*, Taposhi Rabeya, Mohammad Asadullah Asad
DOI: 10.22607/IJACS.2018.604005
Volume 6, Issue 4 | Pages: 200-204
Abstract
The removal of methylene blue (MB) from wastewater was studied using ferric oxide (Fe2O3) as an adsorbent. The adsorption
potential of Fe2O3 was investigated in batch adsorption experiment. The adsorption experiments were carried out with variation
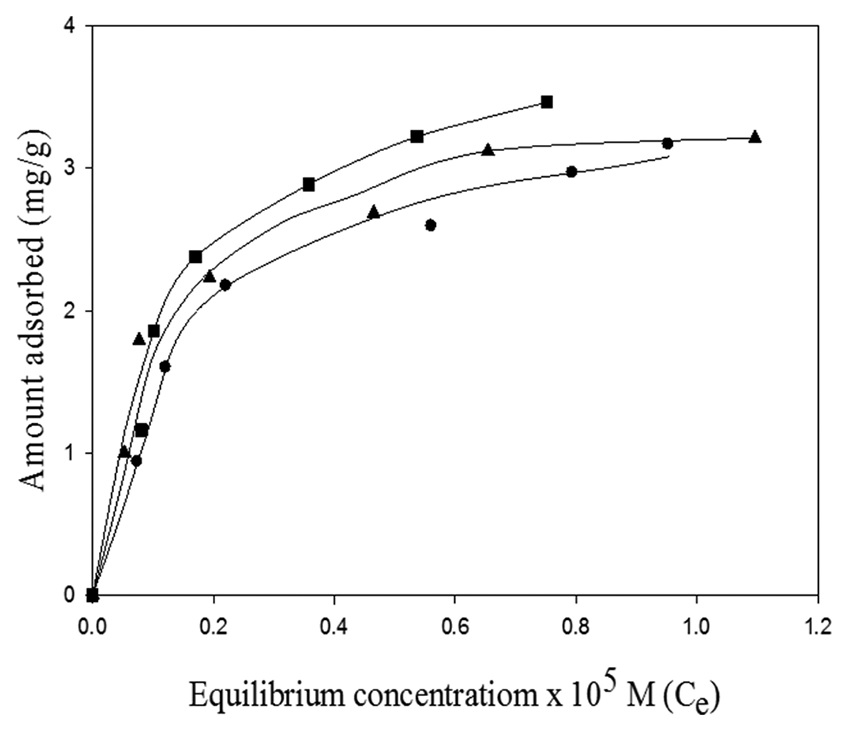
in contact time, initial pH, and temperature of the solution. The equilibrium time for the adsorption of MB on Fe2O3 was found
to be about 3 h. Adsorption isotherms were measured at three different temperatures and found to fit Langmuir isotherm. The
positive values of estimated enthalpy of adsorption were indicative of activated adsorption. Batch adsorption models, based on
the assumptions of the Lagergren pseudo-first-order and the pseudo-second-order model, were applied to predict the kinetics of
the adsorption and data fitted very well to the pseudo-second-order kinetic model.
Keywords
Adsorption Adsorbent Equilibrium Langmuir isotherm.References
No references available for this article.
Citation
Ajoy Kanti Mondal*, Taposhi Rabeya, Mohammad Asadullah Asad. Removal of methylene blue from wastewater using Fe2O3 as an adsorbent. Indian J. Adv. Chem. Sci. 2019; 6(4):200-204.