Synthesis of 4-Aminoantipyrine Derivatives: A Rapid and Eco-Friendly Approach
Kaushik A. Mestry, Dattatray B. Yedage, Chetan B. Meher, Siddheshwar D. Jadhav, Madhuri M. Varma, Dattatray V. Patil
DOI: DOI: 10.22607/IJACS.2025.1302002
Volume 13, Issue 2 | Pages: 73-76
Abstract
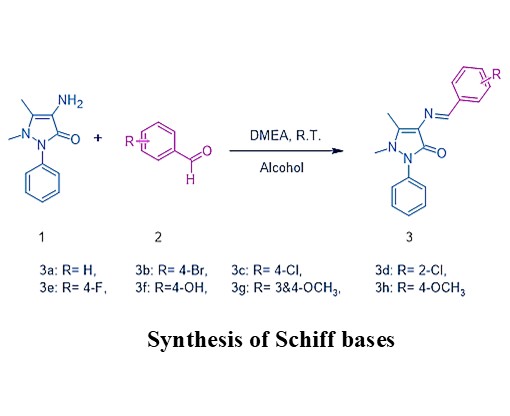
This study presents an eco-friendly and efficient method for synthesizing 4-aminoantipyrine derivatives through a reaction
with aldehydes. Ethanol is employed as a co-solvent, ensuring a sustainable medium, while N,N-dimethylethanolamine acts
as a bifunctional organic catalyst, enhancing reaction efficiency. The reaction proceeds efficiently at ambient temperature,
thereby minimizing energy usage. This method offers high yields in a short time, making it both cost-effective and practical.
The synthesized derivatives were characterized using various spectroscopic techniques to confirm their structural integrity and
purity. This green synthetic approach minimizes the use of hazardous reagents and aligns with sustainable chemistry principles,
demonstrating its potential for environmentally benign organic transformations.
Keywords
N N-Dimethylethanolamine (DMEA) 4-aminoantipyrine Schiff Bases Green synthesis.References
No references available for this article.
Citation
Kaushik A. Mestry, Dattatray B. Yedage, Chetan B. Meher, Siddheshwar D. Jadhav, Madhuri M. Varma, Dattatray V. Patil. Synthesis of 4-Aminoantipyrine Derivatives: A Rapid and Eco-Friendly Approach. Indian J. Adv. Chem. Sci. 2025; 13(2):73-76.