Cyclic Voltammetric Studies of 3,5-Dichloro-2-Hydroxyacetophenone Sulfamethazine -Based Azomethine Compound and its Cu (II) Complex at Glassy Carbon Electrode
Ishwar Chand Balaee*, Gopal Lal Kumawat
DOI: DOI: 10.22607/IJACS.2025.1302005
Volume 13, Issue 2 | Pages: 91-97
Abstract
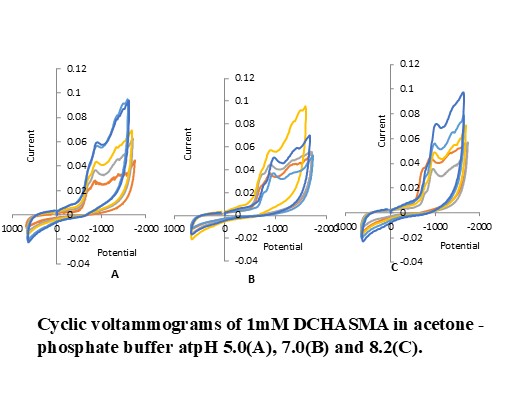
Cyclic voltammetry has been used as an electrochemical method to study the electrochemical redox behavior of 3,5-dichloro-2-hydroxyacetophenone Sulphamethazine (DCHASMA) and its Cu(II) complex. Cyclic voltammograms of DCHASMA were recorded at glassy carbon electrode (GCE) at different pH values using Britton-Robinson (BR) buffer and aqueous phosphate buffer. Under the examined experimental conditions, the complex exhibits a two-electron quasi-reversible reduction wave in the cathodic area, whereas the cyclic voltammograms of DCHASMA consist of an irreversible cathodic wave that follows a one-step, two-electron transfer electrochemical process. Diffusion controlled mechanism was proven by the linear relationship between peak current and the square root of scan rate. On cyclic voltammetric parameters, the effects of solvent, buffer medium, scan rates, and pH were examined. The various kinetic parameters such as charge-transfer coefficient (αn), diffusion coefficient (D01/2) and rate constant (K0f,h) were also calculated.
Keywords
Cyclic Voltammetry 3 5-dichloro-2-hydroxyacetophenone Sulphamethazine(DCHASMA) Charge-Transfer Coefficient (αn) Diffusion coefficient (D01/2) and Rate Constant (K0f h)References
No references available for this article.
Citation
Ishwar Chand Balaee*, Gopal Lal Kumawat. Cyclic Voltammetric Studies of 3,5-Dichloro-2-Hydroxyacetophenone Sulfamethazine -Based Azomethine Compound and its Cu (II) Complex at Glassy Carbon Electrode. Indian J. Adv. Chem. Sci. 2025; 13(2): 91-97.