Thermally Stable and Soluble Polyesters Derived from Naphthyl-Benzamide Diol: Synthesis and Characterization
Shivaji D. Ghodke, Arati V. Diwate, Noormahmad N. Maldar
DOI: DOI: 10.22607/IJACS.2025.1303004
Volume 13, Issue 3 | Pages: 133-139
Abstract
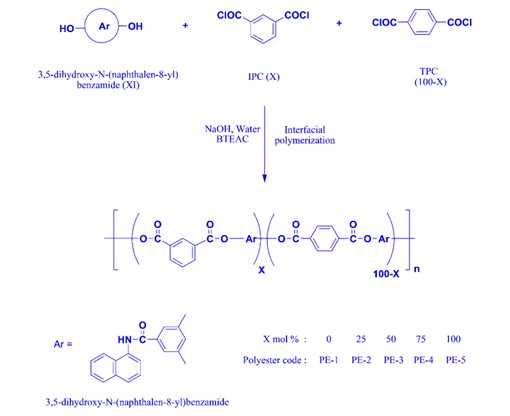
A novel diol, 3,5-dihydroxy-N-(naphthalen-8-yl)benzamide, was successfully synthesized via Yamazaki condensation between 1-naphthylamine and 3,5-dihydroxybenzoic acid in the presence of triphenyl phosphate and pyridine. The structure of the synthesized diol was confirmed through FT-IR, ¹H NMR, and ¹³C NMR spectroscopy. This diol was subsequently polymerized with varying molar ratios of terephthaloyl chloride (TPC) and isophthaloyl chloride (IPC) to produce a series of polyesters (PE-1 to PE-5) using a phase transfer catalyst system. The resulting polyesters were obtained in good yields, with inherent viscosities ranging from 0.40 to 0.88 dL/g. FT-IR analysis confirmed the successful formation of ester linkages. The polyesters demonstrated excellent solubility in polar organic solvents, attributed to the presence of bulky naphthalene pendant groups and polar amide linkages. Wide-angle X-ray diffraction (WAXD) patterns indicated low crystallinity. Thermal analysis via TGA and DSC revealed that the polyesters possessed high thermal stability, with decomposition temperatures reaching up to 488 °C and glass transition temperatures (Tg) in the range of 123–179 °C. These findings suggest that the integration of naphthalene and benzamide units significantly improves the thermal properties and solubility of the polyesters, making them promising candidates for high-performance material applications.
Keywords
Aromatic Polyesters; Naphthalene-Based Diols; Yamazaki Condensation; Thermal Stability; Solubility; Phase Transfer Catalysis; FT-IR Spectroscopy; NMR SpectroscopyReferences
No references available for this article.
Citation
Shivaji D. Ghodke, Arati V. Diwate, Noormahmad N. Maldar. Thermally Stable and Soluble Polyesters Derived from Naphthyl-Benzamide Diol: Synthesis and Characterization. Indian J. Adv. Chem. Sci. 2025; 13(3):133-139.