Proton Affinities of a Series of α, β Unsaturated Carbonyl Compounds of Type-2-alkene (Acrolein, 4-hydroxy-2-nonenal, Methyl Vinyl Ketone, Acrylamide, Methyl Acrylate, and Ethylmethacrylate), in the Gas and Aqueous Phase in their Low-lying Excited Triplet State: A Density Functional Theory/Polarizable Continuum Model and Self-Consistent Reaction Field Approach
Biswarup Mandal, Umasankar Senapati, Bhudeb Ranjan De*
DOI: 10.22607/IJACS.2017.501010
Volume 5, Issue 1 | Pages: 65-75
Abstract
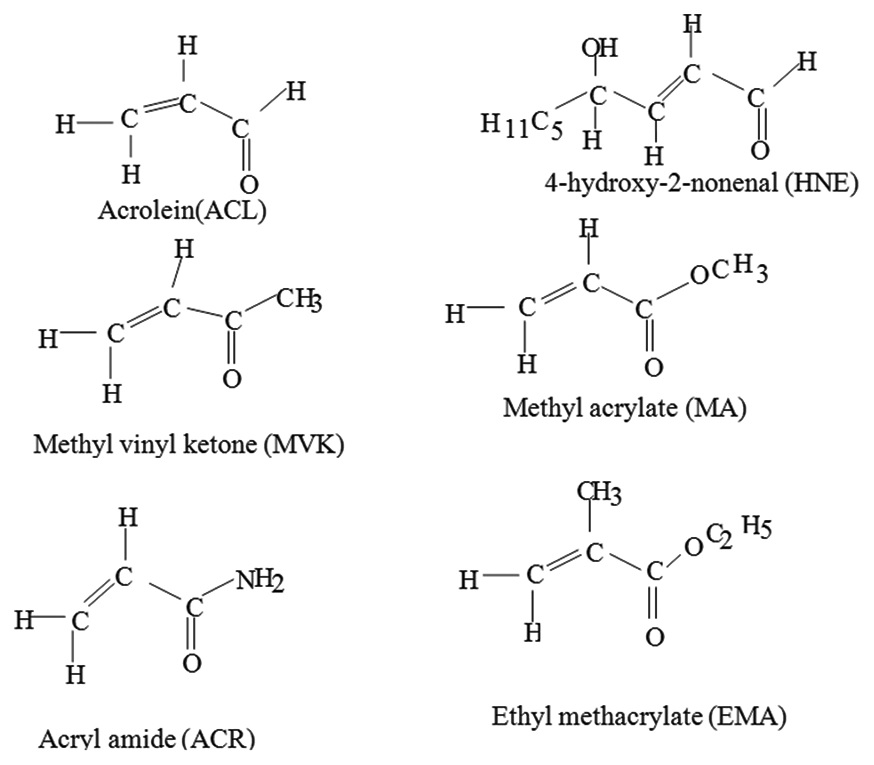
Density functional theory Becke 3-term functional; Lee, Yang, Parr/6-311G(d,p) calculations were performed
to quantify triplet state proton affinities (PA) and transition energies of a series of α,β-unsaturated carbonyl
compounds and their O-protonated counterparts in gas phase as well as in aqueous phase. To evaluate structural
behavior and different quantum mechanical properties in water, we studied our optimization process using
polarizable continuum model and Self-consistent reaction field method at the same level of theory of the relevant
low-lying excited state. The gas phase O-protonation turns out to be exothermic in each case and the local
stereochemical disposition of the proton is found to be almost the same in each case. PA values of the different
compounds are affected by substituent present at the carbonyl carbon. Different electrochemical properties
(+R, +I, and effect) originate from carbonyl chain are seen to cause change of the PAs. Acrylamide (ACR) shows
the highest PA in both phases. In each case, protonation at carbonyl oxygen is observed to be more energetically
favorable compared to protonation at other probable binding sites present. Computed PAs of the compounds in
gas phase are in the following order ACR ≥ ethylmethacrylate > 4-hydroxy-2-nonenal > methyl vinyl ketone
> methyl acrylate > acrolein, while in aqueous phase the PA order is ranked differently. Charge density on
binding oxygen and on added proton is recorded from both Mulliken population analysis and Natural population
analysis. PA values are sought to be correlated with the computed hardness of the unprotonated species in the
relevant excited state. The proton induced shifts are in general red shifts for the low-lying excited triplet state.
The overall reactivity is explained by distant atom contribution in addition to the contribution from the carbonyl
group.
Keywords
Density functional theory Polarisable continuum model Proton affinity Low-lying Unsaturated.References
No references available for this article.
Citation
Biswarup Mandal, Umasankar Senapati, Bhudeb Ranjan De*. Proton Affinities of a Series of α, β Unsaturated Carbonyl Compounds of Type-2-alkene (Acrolein, 4-hydroxy-2-nonenal, Methyl Vinyl Ketone, Acrylamide, Methyl Acrylate, and Ethylmethacrylate), in the Gas and Aqueous Phase in their Low-lying Excited Triplet State: A Density Functional Theory/Polarizable Continuum Model and Self-Consistent Reaction Field Approach. Indian J. Adv. Chem. Sci. 2017; 5(1):65-75.