Supramolecular Assembly of Ionic Liquid with Cyclic Polyethers to Form Inclusion Complexes
Mitali Kundu, Mahendra Nath Roy*
DOI: 10.22607/IJACS.2017.502004
Volume 5, Issue 2 | Pages: 92-101
Abstract
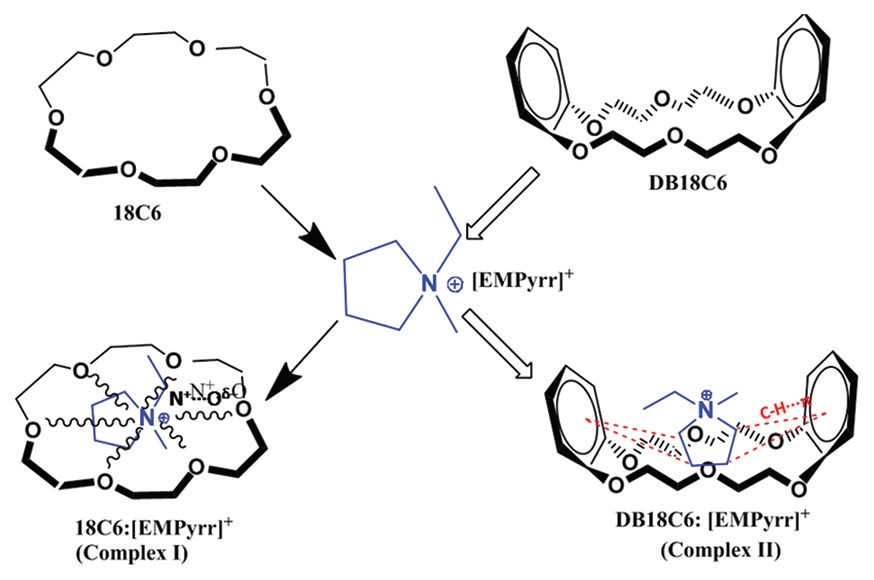
Complexation process of the selected macrocyclic crown compound such as 18-crown-6 (18C6) and dibenzo 18C6
(DB18C6) with 1‑ethyl‑1‑methylpyrrolidinium hexafluorophosphate [EMPyrr] PF6 as a guest molecule have
been intended in acetonitrile medium at by conductometry (three different temperatures), 1H nuclear magnetic
resonance (NMR) and mass spectra. The formation constants of the complexes are evaluated in solution phase
from the molar conductance-mole ratio values. The result shows that the stability of the resulting complexes with
the same inward cationic moiety follows in the order 18C6: [EMPyrr]+>DB18C6:[EMPyrr]+ and based on the
character of the crown ethers. The calculated thermodynamic parameters support this complexation process.
The host‑guest complexations of the five‑membered nitrogen‑containing cation with two different macrocyclic
polyethers are supported by studying 1H NMR. The electrospray mass spectrometry has been used to support the
complexation process with the proper stoichiometry ratio. The solid complex formation between the selected two
crown ethers and the ionic liquid are established by Fourier transform infrared study.
Keywords
Formation constant 1H nuclear magnetic resonance Electrospray ionization mass spectrometry Fourier transform infrared spectroscopy.References
No references available for this article.
Citation
Mitali Kundu, Mahendra Nath Roy*. Supramolecular Assembly of Ionic Liquid with Cyclic Polyethers to Form Inclusion Complexes. Indian J. Adv. Chem. Sci. 2017; 5(2):92-101.