Investigation on Solvation Behavior of an Ionic Liquid (1-butyl-3-methyl imidazolium Chloride) with the Manifestation of Ion Association Prevailing in Different Pure Solvent Systems
Siti Barman, Biswajit Datta, Mahendra Nath Roy*
DOI: 10.22607/IJACS.2017.503008
Volume 5, Issue 3 | Pages: 160-175
Abstract
The ion-pair formation constant (KP) and triple-ion formation constant (KT) of 1-butyl-3-methylimidazolium
chloride ([bmim][Cl]) have been determined conductometrically in different solvent media in the temperature
range from 298.15 to 318.15 K. The Fuoss conductance equation (1978) for ion-formation and Fuoss–Kraus
theory for triple-ion formations have been used for analyzing the conductance data. The Walden product is
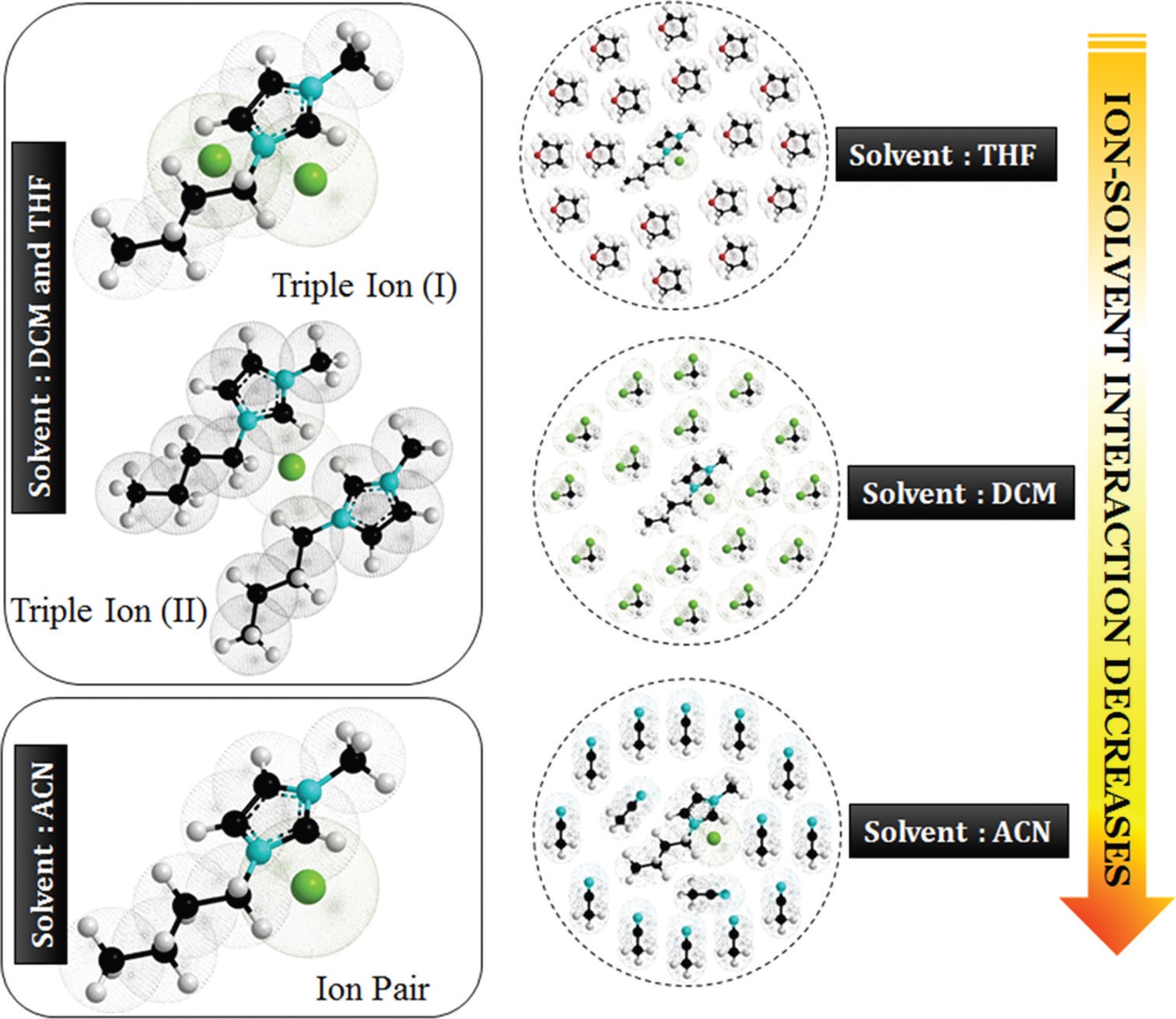
obtained and discussed. However, the deviation of the conductometric curves (Λ vs. √m) from linearity for the
electrolyte in tetrahydrofuran and dichloromethane indicated/indicates triple-ion formation. Ion-solvent
interactions have been studied with the help of density, viscosity, and Fourier transform infrared spectroscopic
measurements. Apparent molar volume and viscosity B-coefficient have been calculated from experimental density
and viscosity data, respectively. The limiting ionic conductances ( o
± ) have been estimated from the appropriate
division of the limiting molar conductance of tetrabutylammonium tetraphenylborate as “reference electrolyte”
method.
Keywords
Ionic liquid Ion-pair and triple-ion formation Ion-solvent interaction Thermodynamic parameters Walden product.References
No references available for this article.
Citation
Siti Barman, Biswajit Datta, Mahendra Nath Roy*. Investigation on Solvation Behavior of an Ionic Liquid (1-butyl-3-methyl imidazolium Chloride) with the Manifestation of Ion Association Prevailing in Different Pure Solvent Systems. Indian J. Adv. Chem. Sci. 2017; 5(3):160-175.