Ultrasonic Studies on the Molecular Interaction of Certain Aliphatic Dialdehyde Compounds with Ethylenediamine in n-Hexane at Different Temperature
Y. Geetha, S. Chidambara Vinayagam*
DOI: 10.22607/IJACS.2017.504002
Volume 5, Issue 4 | Pages: 220-229
Abstract
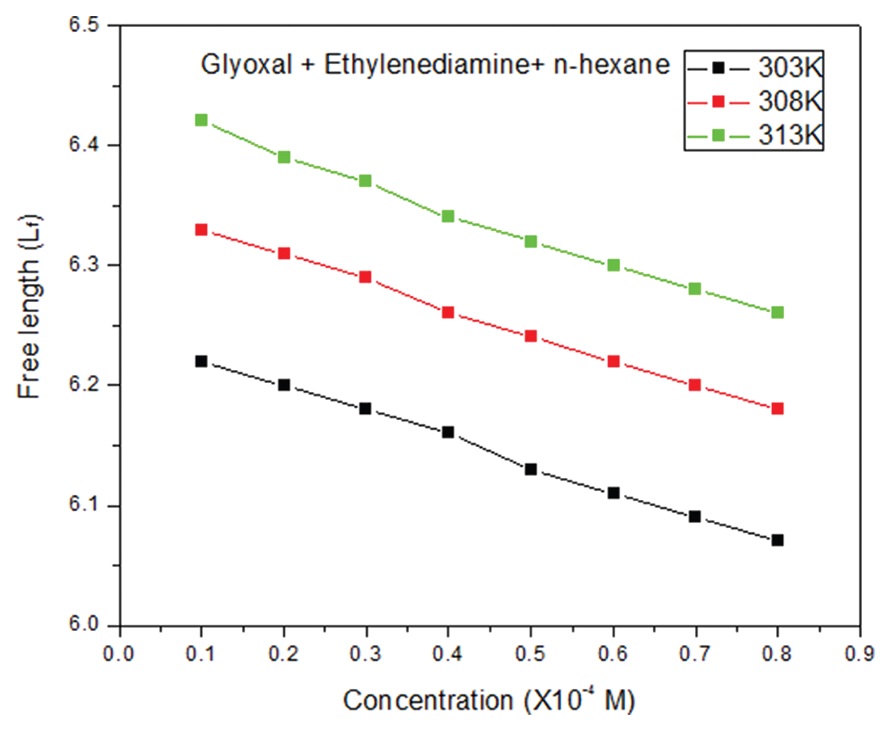
Acoustical parameters are calculated for three ternary systems of ethylenediamine and different dialdehydes
such as glyoxal, glutaraldehyde, and succinaldehyde in n-hexane medium from the measurement of ultrasonic
velocity, density, and viscosity at 303K, 308K, and 313K. From these data, acoustical parameters such as
adiabatic compressibility, free length, free volume, internal pressure, absorption coefficient, viscous relaxation
time, available volume, cohesive energy, Lenard-Jones potential, free energy of activation, formation constant,
and molecular interaction parameter have been evaluated. From the measurements, investigated the complex
formation through intermolecular hydrogen bonding between dialdehyde and ethylenediamine. Excess thermo
acoustic parameters conclude the existence of hydrogen-bonded complexes between dialdehyde and amine.
Keywords
Dialdehyde Ethylenediamine Intermolecular hydrogen bond Excess parameter.References
No references available for this article.
Citation
Y. Geetha, S. Chidambara Vinayagam*. Ultrasonic Studies on the Molecular Interaction of Certain Aliphatic Dialdehyde Compounds with Ethylenediamine in n-Hexane at Different Temperature. Indian J. Adv. Chem. Sci. 2018; 5(4):220-229 .