Solute-solute and Solute-solvent Interactions of Paracetamol in Aqueous Solutions of β-cyclodextrin at Different Temperatures: A Volumetric and Viscometric Approach
Abhijit Sarkar, Habibur Rahaman, Uttam Kumar Singha, Biswajit Sinha*
DOI: 10.22607/IJACS.2017.504003
Volume 5, Issue 4 | Pages: 230-244
Abstract
The densities and viscosities of paracetamol in aqueous b-cyclodextrin solutions with several molal concentrations
m = (0.001-0.007) mol kg−1 of b-cyclodextrin were determined at T = 298.15-318.15 K under atmospheric
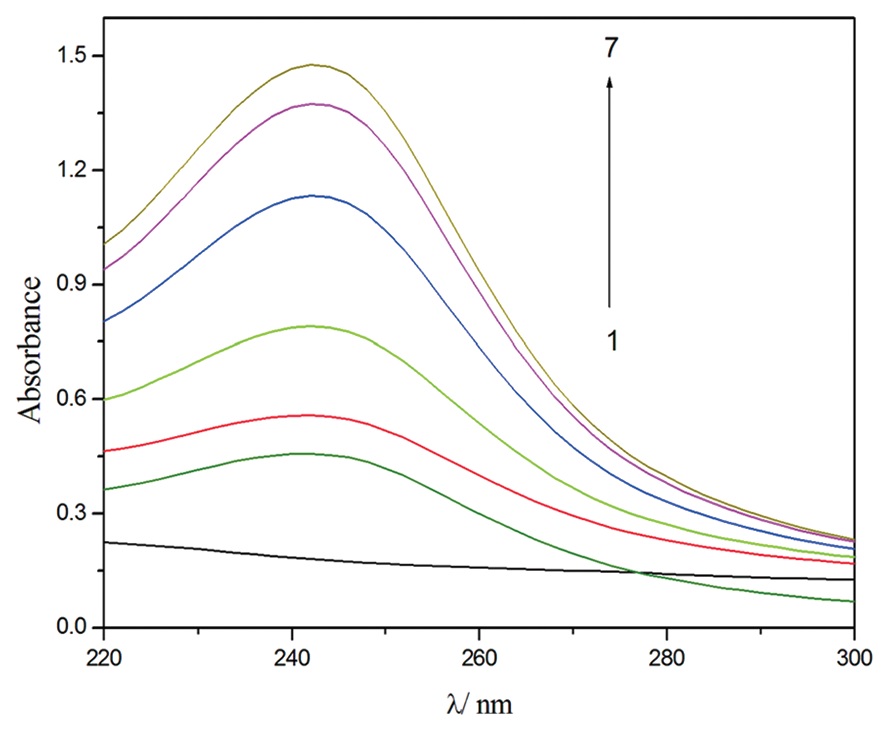
pressure. The inclusion has been studied using ultraviolet-visible spectroscopy. Using experimental data
apparent molar volume (φV), standard partial molar volume ( 0
Vφ ), the slope ( SV
* ), standard isobaric partial
molar expansibility ( φE
0 ) and its temperature dependence (∂φ ∂E
0
PT ) , the viscosity B-coefficient, and solvation
number (Sn) were determined. Free energies of activation of viscous flow per mole of the solvents ( ∆μ ≠
1
0 ) and the
solute ( ∆μ ≠
2
0 ) are also calculated. Various results revealed that the solutions are characterized predominantly by
solute-solvent interactions and paracetamol behaves as a long-range structure maker.
Keywords
Apparent molar volumes Viscosity B-coefficients Paracetamol Aqueous b-cyclodextrin solutions Solvation number Ultraviolet-visible spectroscopy.References
No references available for this article.
Citation
Abhijit Sarkar, Habibur Rahaman, Uttam Kumar Singha, Biswajit Sinha*. Solute-solute and Solute-solvent Interactions of Paracetamol in Aqueous Solutions of β-cyclodextrin at Different Temperatures: A Volumetric and Viscometric Approach. Indian J. Adv. Chem. Sci. 2018; 5(4):230-244 .