Investigation of Diverse Interactions of Lithium Hexafluoroarsenate Prevailing in Pure and Mixed Industrial Solvent Systems by Physicochemical Methodology
Biraj Kumar Barman, Deepak Ekka, Rajani Dewan, Mahendra Nath Roy*
DOI: 10.22607/IJACS.2017.504009
Volume 5, Issue 4 | Pages: 285-294
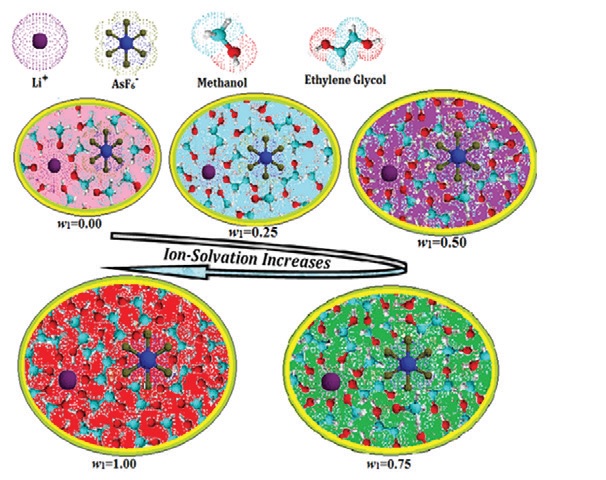
Abstract
Precise measurements of electrolytic conductivities, densities, and viscosities of lithium hexafluoroarsenate
(LiAsF6) have been reported in w1=0.00, 0.25, 0.50, 0.75, 1.00 mass fraction of ethylene glycol in methanol at
298.15 K. The limiting molar conductivities (Λ0), association constants (KA), and the distance of closest approach
of the ions (R) have been evaluated using the Fuoss conductance equation (1978) for ion-pair formation. The
Walden products have been obtained and discussed. Apparent molar volumes (fV) and viscosity B-coefficients
have been determined from the density (ρ) and viscosity (h) data, respectively. The limiting apparent molar
volumes (fVo), and experimental slopes (SV*) have been obtained from Masson equation and interpreted in terms
of ion-solvent and ion-ion interactions, respectively. The viscosity data have been analyzed using Jones-Dole
equation, and the derived parameters A- and B-coefficient have also been interpreted for ion-ion and ion-solvent
interactions, respectively, in the solutions.
Keywords
Lithium hexafluoroarsenate Ethylene glycol Methanol Conductivity Density Viscosity.References
No references available for this article.
Citation
Biraj Kumar Barman, Deepak Ekka, Rajani Dewan, Mahendra Nath Roy*. Investigation of Diverse Interactions of Lithium Hexafluoroarsenate Prevailing in Pure and Mixed Industrial Solvent Systems by Physicochemical Methodology. Indian J. Adv. Chem. Sci. 2018; 5(4):285-294 .