Synthesis, Spectroscopic and Thermal Studies of Fe(III) and VO(IV) Complexes of Heterocyclic Schiff Base Ligand
A. P. Thakare*, P. R. Mandlik
DOI: 10.22607/IJACS.2017.504013
Volume 5, Issue 4 | Pages: 318-323
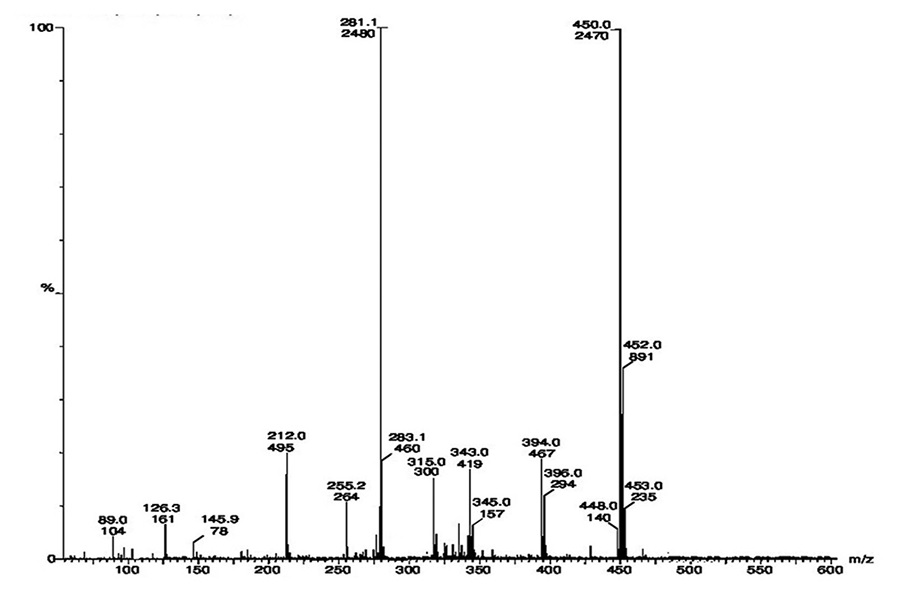
Abstract
The present article describes the synthesis, characterization and thermal studies of newly synthesized Fe(III)
and VO(IV) complexes from heterocyclic Schiff base ligand. The new Schiff base ligand has been synthesized
by refluxing chalcone 3-chloro-6-hydroxy-2-methylphenyl-3-(3,4-dimethoxyphenyl)prop-2-en-1-one with
isonicotinic hydrazide in the ethanolic medium. The reaction of this ligand with metal salts results in the
formation of respective metal complexes. The compounds were characterized on the basis of elemental analysis,
molar conductance, infrared (IR), ultraviolet-visible, mass, and 1H-nuclear magnetic resonance spectroscopic
techniques. IR spectral data show that ligand act as monobasic tridentate ONO donor toward Fe(III) ion and
monobasic bidentate ON donor toward VO(IV) ion. Thermal stability and decomposition behavior of complexes
have been studied with the help of thermogravimetric technique at a heating rate 10°C min−1 in a nitrogen
atmosphere.
Keywords
Schiff base Metal complexes Infrared Thermal analysis.References
No references available for this article.
Citation
A. P. Thakare*, P. R. Mandlik. Synthesis, Spectroscopic and Thermal Studies of Fe(III) and VO(IV) Complexes of Heterocyclic Schiff Base Ligand. Indian J. Adv. Chem. Sci. 2018; 5(4):318-323 .