Conductivity and Dielectric Behavior of Polyethylene Oxide-Lithium Perchlorate Solid Polymer Electrolyte Films
J. Gurusiddappa, W. Madhuri*, R. Padma Suvarna, K. Priya Dasan
DOI:
Volume 4, Issue 1 | Pages: 14-19
Abstract
Solid polymer electrolytes (SPEs) consisting of polyethylene oxide (PEO) complexed with lithium perchlorate
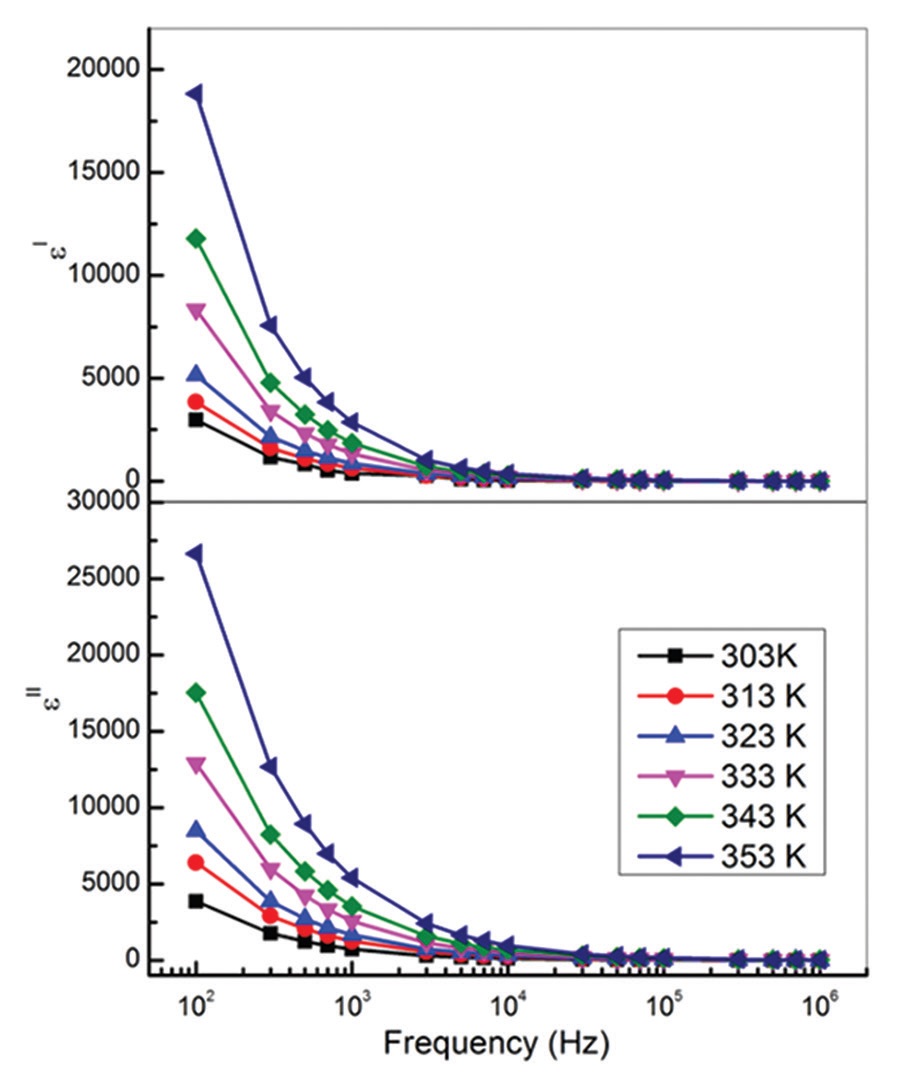
(LiClO4) have been synthesized by solution casting method. The ionic conductivity, dielectric response of the SPE
system is studied within the frequency range of 100 Hz - 1 MHz and in the temperature range of 303-353 K. The
room temperature ionic conductivity value of PEO-LiClO4 electrolyte is found to be ~10−5 S cm−1. The dielectric
properties were studied using the complex dielectric permittivity spectra and complex electric modulus spectra of
the SPE films. It is observed that magnitude of dielectric permittivity is high in the lower frequency region due to
electrode polarization (EP) effect. The dielectric permittivity and loss were increased with increase of temperature.
The AC conductivity follows universal power law with a small deviation in the EP dominated low frequency region.
Keywords
Solid polymer electrolytes Polyethylene oxide Lithium perchlorate and dielectric.References
No references available for this article.
Citation
J. Gurusiddappa, W. Madhuri*, R. Padma Suvarna, K. Priya Dasan. Conductivity and Dielectric Behavior of Polyethylene Oxide-Lithium Perchlorate Solid Polymer Electrolyte Films. Indian J. Adv. Chem. Sci. 2016; 4(1):14-19.