Theoretical Approach on Structural Aspects of a Potent, Selective, Orally Bioavailable Hedgehog Antagonist, 2-chloro-N-[4-chloro-3-(pyridin-2-yl)phenyl]-4-(methylsulfonyl) benzamide
Amaku Friday James*, Otuokere Ifeanyi Edozie
DOI:
Volume 4, Issue 1 | Pages: 31-35
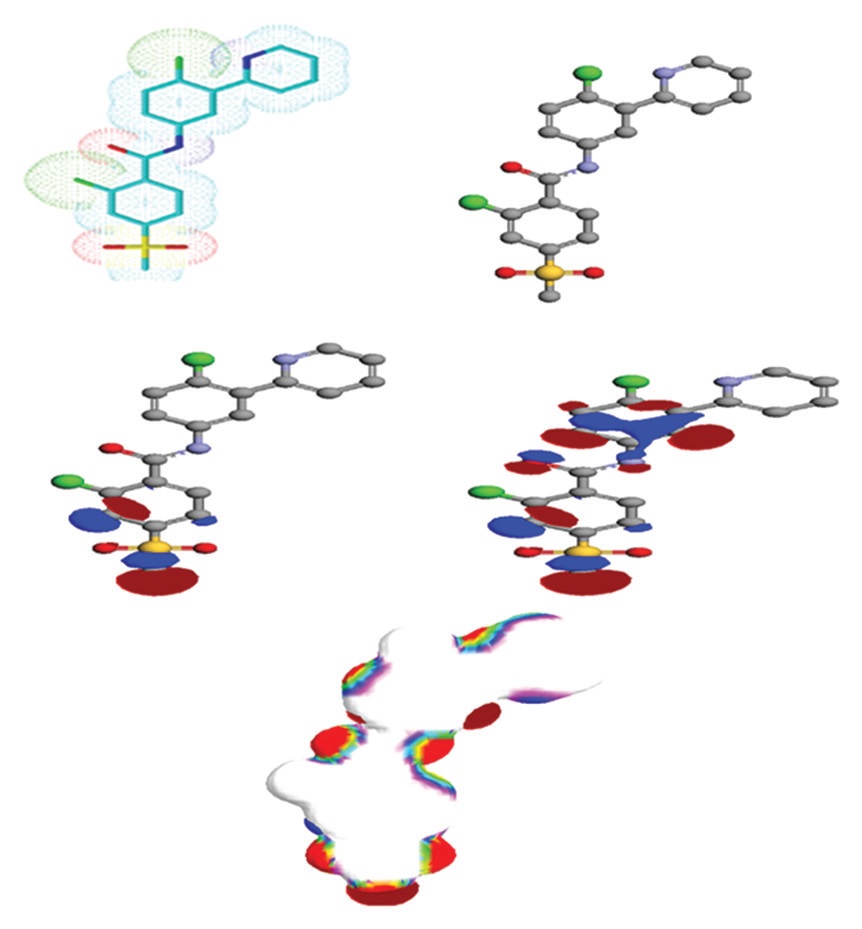
Abstract
The molecular mechanics potential energy function were evaluated in terms of energies associated with bonded
interactions (bond length, bond angle, and dihedral angle) as well as non-bonded interactions (Vander Waals
and electrostatic). Surfaces were created to visualize excited state properties such as highest occupied molecular
orbitals, lowest unoccupied molecular orbitals, and electrostatic potential mapped density. The steric energy
for 2-chloro-N-[4-chloro-3-(pyridin-2-yl)phenyl]-4-(methylsulfonyl)benzamide (vismodegib) was calculated to
be 0.54948080 au (344.80472191 kcal/mol). The most energetically favorable conformation of vismodegib was
found to have a heat of formation of 1585.2455 kcal/mol. The self-consistent field energy was calculated by
geometry convergence function using RHF/AM1 method with a net charge of 0 and valence electron of 124, in
ArgusLab software. The most feasible position for vismodegib to act as a highly potent and totally selective orally
bioavailable Hedgehog antagonist was found to be −171.973538 au (−107915.121800 kcal mol−1).
Keywords
Vismodegib Molecular mechanics ArgusLab software.References
No references available for this article.
Citation
Amaku Friday James*, Otuokere Ifeanyi Edozie. Theoretical Approach on Structural Aspects of a Potent, Selective, Orally Bioavailable Hedgehog Antagonist, 2-chloro-N-[4-chloro-3-(pyridin-2-yl)phenyl]-4-(methylsulfonyl) benzamide. Indian J. Adv. Chem. Sci. 2016; 4(1):31-35.