Ultraviolet Light Induced Oxidation of Malonic Acid by Chloramine-T in an Aqueous Acidic Medium: A Kinetic Study
Meena Wadhwani*, Shubha Jain
DOI:
Volume 4, Issue 1 | Pages: 36-39
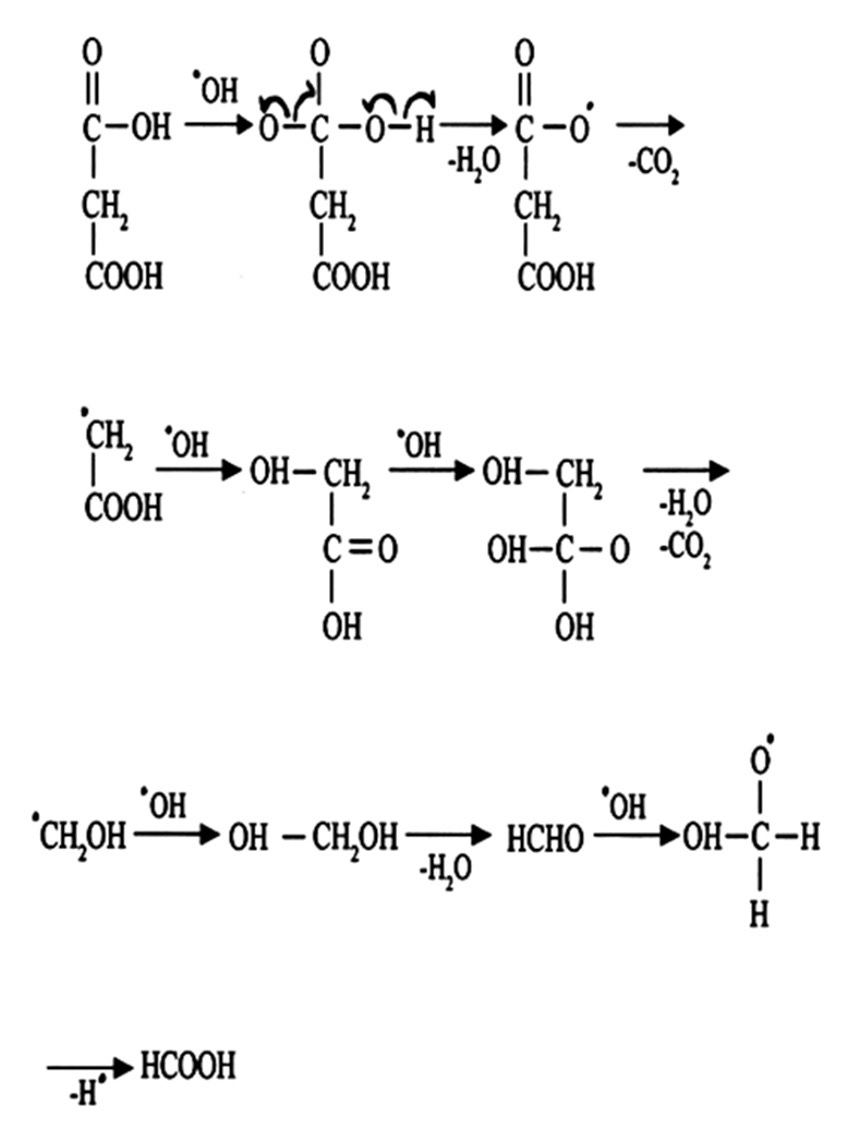
Abstract
The kinetics of the photo-oxidation of malonic acid by chloramine-T in an aqueous medium has been studied. It is
found that reaction obeys first order kinetics with respect to each of the substrate as well as oxidant. The reaction
is found to be catalyzed by H+ ions. Different parameters such as the effect of the variation of concentration of the
reactants, product, and intensity of light on the reaction rate have been studied. A suitable mechanism has been
proposed depending on the experimental findings.
Keywords
Photo-oxidation Chloramine-T Malonic acid Hydrochloric acid.References
No references available for this article.
Citation
Meena Wadhwani*, Shubha Jain. Ultraviolet Light Induced Oxidation of Malonic Acid by Chloramine-T in an Aqueous Acidic Medium: A Kinetic Study. Indian J. Adv. Chem. Sci. 2016; 4(1):36-39.