Spectrophotometric Determination of Tiemonium Methyl Sulfate, Itopride Hydrochloride and Trimebutine Maleate via Ion Pair Complex Formation and Oxidation Reaction
M. Ayad, M. El-Balkiny, M. Hosny*, Y. Metias
DOI:
Volume 4, Issue 1 | Pages: 85-97
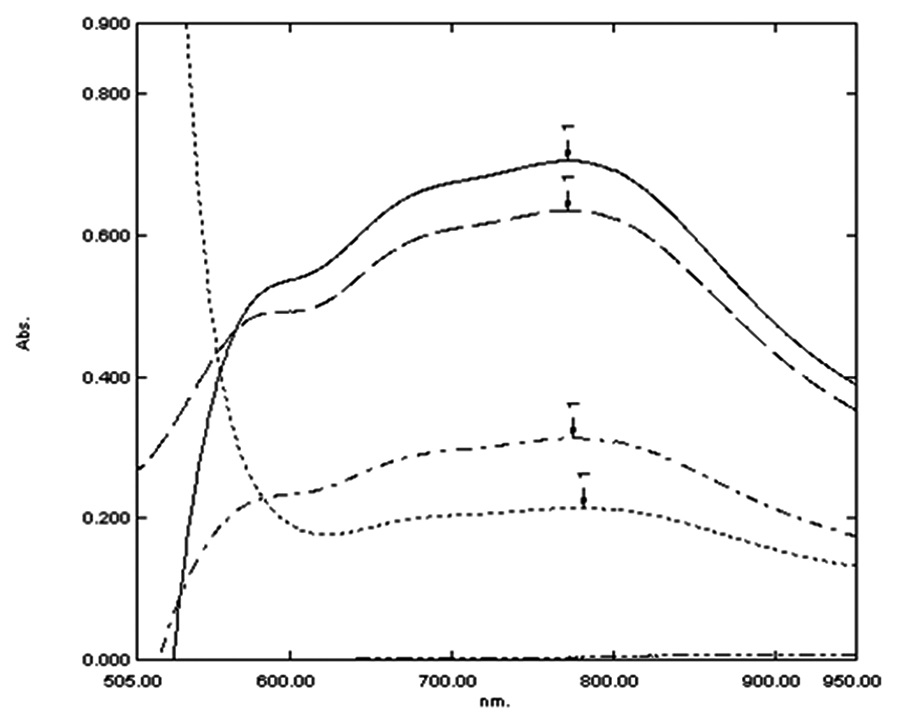
Abstract
Two simple and precise spectrophotometric methods were developed for quantitative determination of
tiemonium methyl sulfate (TIM), itopride hydrochloride (ITH), and trimebutine maleate (TRM) in both pure and
pharmaceutical dosage forms. Method A: Based on oxidation of the studied drugs with ammonium vanadate
in sulfuric acid medium resulting in greenish blue color measured at 773 nm for TIM, ITH and TRM. Method
B: Involved formation of an ion pair complex between the studied drugs and rose bengal which were measured
at 569, 573, 565 nm for TIM, ITH, and TRM, respectively in acetate buffered medium of pH 3.7 using methyl
cellulose as a surfactant. The effect of different variables on the involved reactions and the color development
were studied and optimized. Beer’s law was obeyed in the range of 20-220 μg ml−1, 3-16 μg ml−1 for TIM,
10-130 μg ml−1, 3-14 μg ml−1 for ITH and 30-170 μg ml−1, 2-12 μg ml−1 for TRM using Methods A and B. Results
of the analysis were validated statistically by recovery studies.
Keywords
Tiemonium Itopride Trimebutine Vanadate Rose bengal.References
No references available for this article.
Citation
M. Ayad, M. El-Balkiny, M. Hosny*, Y. Metias . Spectrophotometric Determination of Tiemonium Methyl Sulfate, Itopride Hydrochloride and Trimebutine Maleate via Ion Pair Complex Formation and Oxidation Reaction. Indian J. Adv. Chem. Sci. 2016; 4(1):85-97.