Ultrasound Assisted Synthesis of Triazole/Tetrazole Hybrids Based New Biquinoline Derivatives as a New Class of Antimicrobial and Antitubercular Agents
Jayvirsinh D. Gohil, Haresh B. Patel, Manish P. Patel*
DOI:
Volume 4, Issue 1 | Pages: 102-113
Abstract
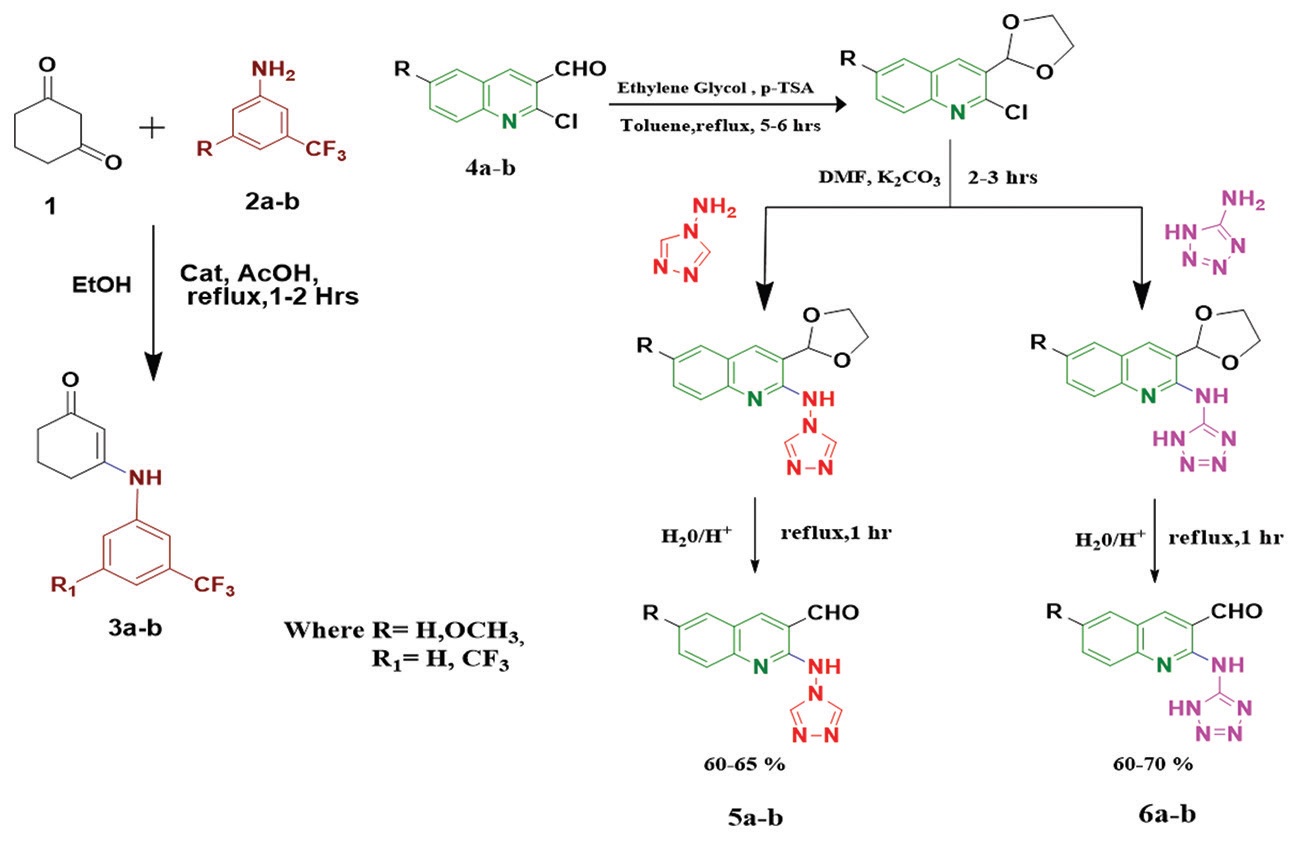
A series of new biquinoline derivatives 8a-h and 9a-h, decorated with 1,2,4-triazole, tetrazole at C-4, and
aromatic trifluoromethyl moiety at N-1 position were synthesized by multicomponent reactions of 2-amino triazole/
amino tetrazole-3-formyl quinoline 5a-b/6a-b with malononitrile/isopropyl cyanoacetate 7a/b and synthesized
β-enaminones 3a/b in the presence of ultrasound (soon-synthetic). The structures of synthesized compounds were
characterized by infrared,1H nuclear magnetic resonance (NMR),13C NMR, and mass spectroscopy. Furthermore,
the synthesized compounds were evaluated for their in vitro antimicrobial activity against a representative panel
of pathogenic strains, anti-tuberculosis (TB) activity against Mycobacterium TB H37Rv.
Keywords
Ultrasound Biquinoline Triazole Tetrazole Antimicrobial Anti-tubercular activity.References
No references available for this article.
Citation
Jayvirsinh D. Gohil, Haresh B. Patel, Manish P. Patel*. Ultrasound Assisted Synthesis of Triazole/Tetrazole Hybrids Based New Biquinoline Derivatives as a New Class of Antimicrobial and Antitubercular Agents. Indian J. Adv. Chem. Sci. 2016; 4(1):102-113.