Study on Solution Behavior of Some Oxalate Salts in Aqueous Vitamin Solutions
Koyeli Das, Mahendra Nath Roy*
DOI:
Volume 4, Issue 2 | Pages: 138-148
Abstract
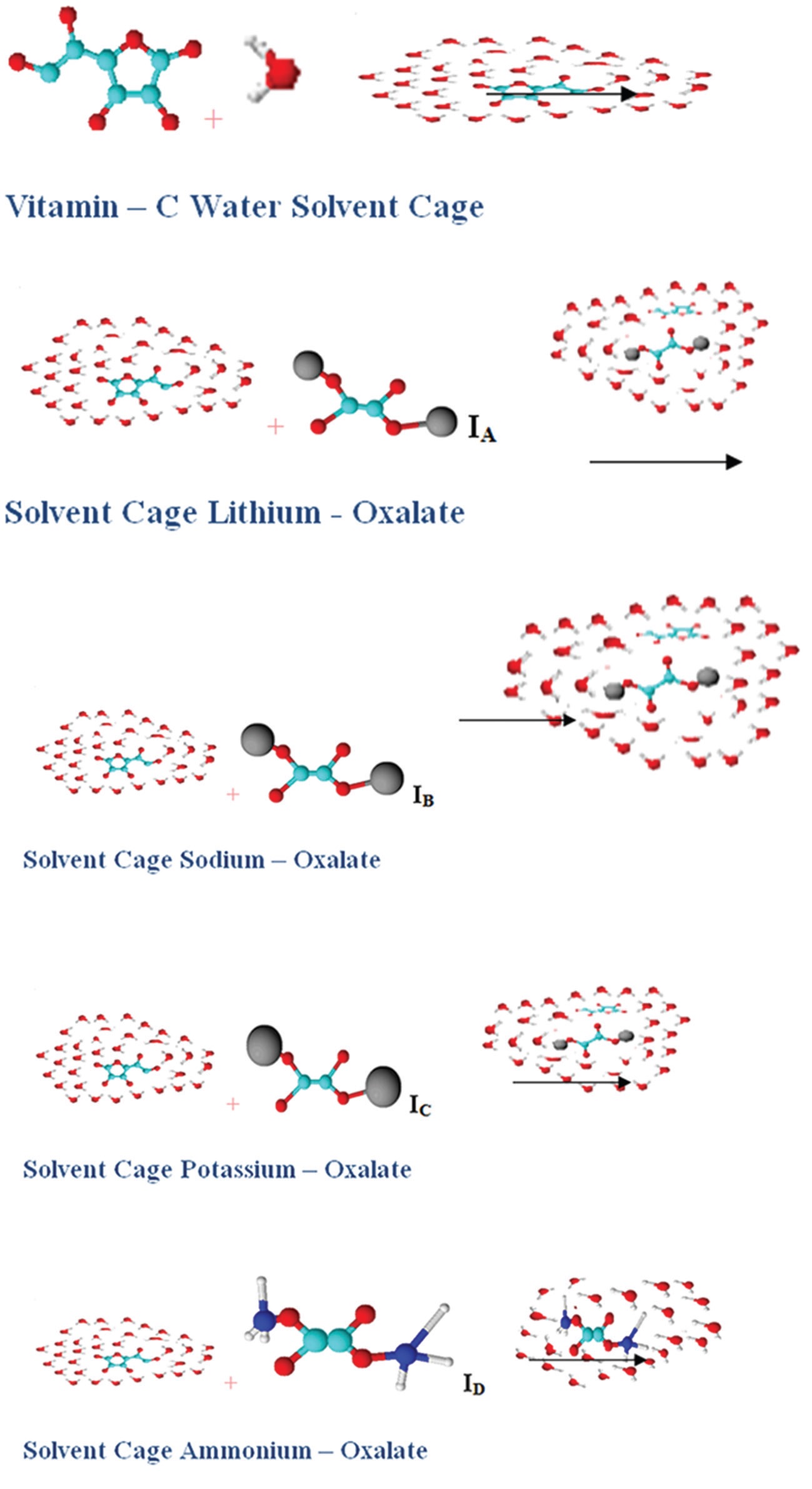
The solubility of solids in vitamin C has much impact in cell metabolism on dissolution in water. Densities
(ρ), viscosities (η), apparent molar volume, refractive index (nD), of oxalate salts (Li2C2O4, Na2C2O4, K2C2O4,
(NH4)2C2O4) have been studied in (0.005, 0.02, 0.035, 0.05, 0.065, 0.08) mass fraction of ascorbic acid in water
(H2O) at 298.15 K, respectively. Masson equation has been employed to find the extent of interaction (solute-
solvent interaction) in terms of the limiting apparent molar volume ( φv
0 ) by extrapolating to zero concentration
and experimental slopes ( SV
* ) which interpreted the solute-solvent and solute-solute interactions, respectively, in
the solutions. Using the Jones–Dole equation, the viscosity data were analyzed to determine the viscosity A and
B-coefficient, which have also been interpreted the solute-solute and solute-solvent interaction, respectively, in
the solutions. Molar refractions (RM) have been calculated with the help of the Lorentz–Lorenz equation. The role
of the solvent and the contribution of solute-solute and solute-solvent interactions to the solution complexes have
also been analyzed through the derived properties. The Gibbs energies of mixing for K2C2O4-vitamin-C binary
solids and liquids and solid-saturated K2C2O4-vitamin-C-H2O ternary liquids were modeled using asymmetric
Margules treatments.
Keywords
Solution chemistry Thermodynamics Apparent molar volume ·Molecular interaction Solute-solute interaction Oxalate salts Ascorbic acid Physico-chemical properties.References
No references available for this article.
Citation
Koyeli Das, Mahendra Nath Roy*. Study on Solution Behavior of Some Oxalate Salts in Aqueous Vitamin Solutions. Indian J. Adv. Chem. Sci. 2016; 4(2):138-148.