Volumetric, Acoustic, and Fourier Transform Infrared Spectroscopic Study of Potassium Thiocyanate in Cyclohexanone, N, N-Dimethylformamide and Dimethyl Sulfoxide
Abhijit Sarkar, Vikas Kumar Dakua, Dipu Kumar Mishra, Biswajit Sinha*
DOI:
Volume 4, Issue 2 | Pages: 180-187
Abstract
Partial molar volumes of potassium thiocyanate (KSCN) in cyclohexanone, N, N-dimethylformamide, and
dimethyl sulfoxide have been determined from solution density measurements at various temperatures and various
electrolyte concentrations. Furthermore, adiabatic compressibility of different solutions has been determined
from the measurement of ultrasonic speeds of sound at 298.15 K. The experimental density data were evaluated by
Masson equation, and the derived parameters were interpreted in terms of ion-solvent and ion-ion interactions.
The structure making or breaking capacity of the electrolyte under investigation has been discussed. Analysis of
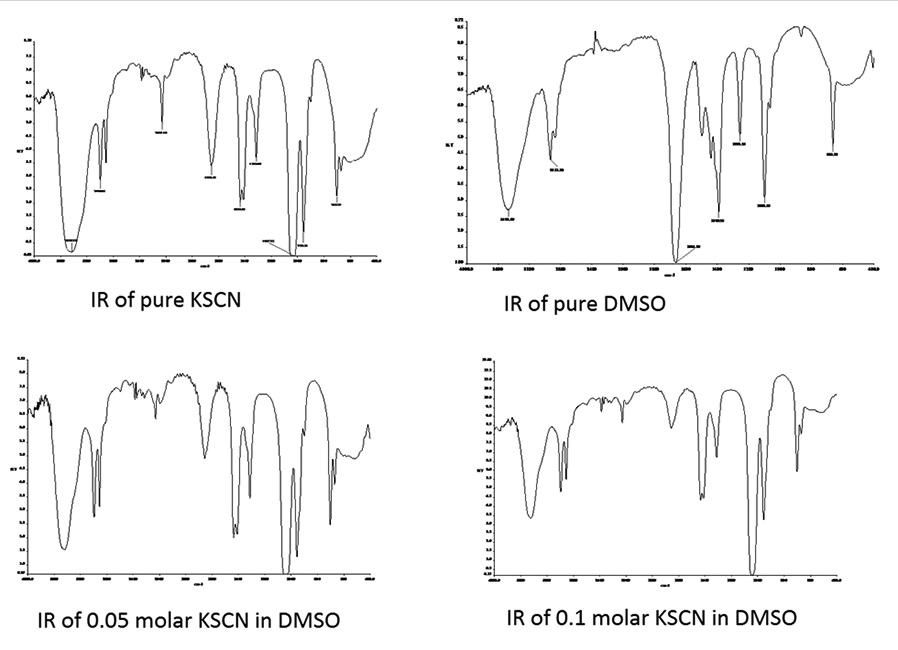
compressibility data indicated the electrostriction of solvent molecules around the metal ion. The infrared spectra
of different solutions at 298.15 K suggest the presence of “spectroscopically free” thiocyanate (CNS−) ion in these
solutions. The cation was found to be substantially solvated in these solvents, whereas the anion appeared to have
a weak interaction with the solvent molecules. For KSCN, the solvent separated ion-pairs predominate over the
contact species, and the observed process has been interpreted by an Eigen multistep mechanism.
Keywords
Partial molar volumes Adiabatic compressibility Ion-solvent and ion-ion interactions Electrostriction Infrared spectra.References
No references available for this article.
Citation
Abhijit Sarkar, Vikas Kumar Dakua, Dipu Kumar Mishra, Biswajit Sinha*. Volumetric, Acoustic, and Fourier Transform Infrared Spectroscopic Study of Potassium Thiocyanate in Cyclohexanone, N, N-Dimethylformamide and Dimethyl Sulfoxide. Indian J. Adv. Chem. Sci. 2016; 4(2):180-187.