Spectrophotometric Determination of Nitrous Acid in the Aqueous Streams of Fast Reactor Fuel Reprocessing by using Azo dye Method
M.Suba, P.Velavendan, N.K. Pandey, C. Mallika and U. Kamachi Mudali
DOI:
Volume 4, Issue 3 | Pages: 321-326
Abstract
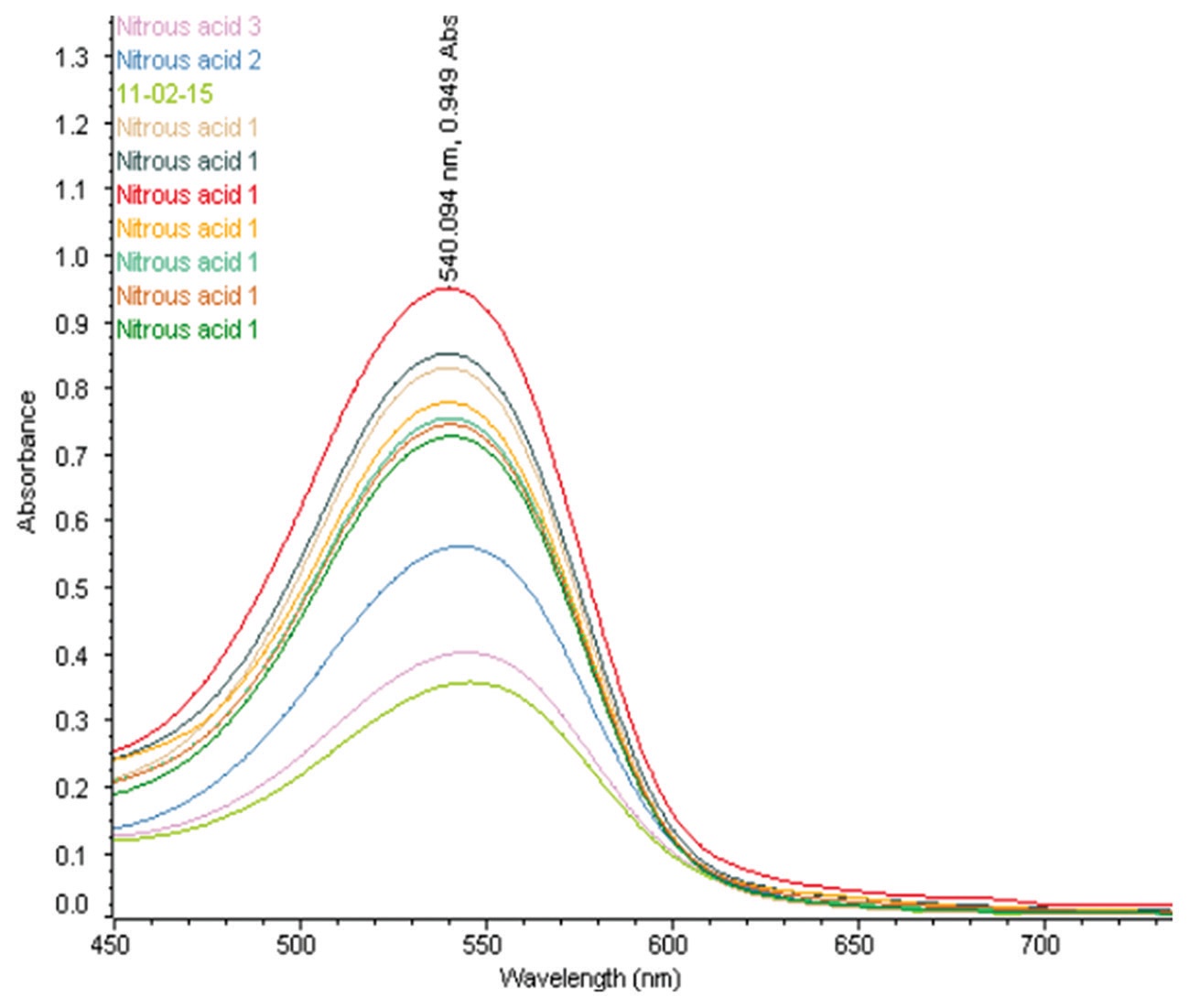
A simple spectrophotometric method is optimized for the determination of nitrous acid in nitric acid media. This
method is based on the diazotization reaction of nitrous acid with sulfanilamide and N-(1-naphthyl)-ethylene
diamine dihydrochloride forming a bright pink colored azo compound. This complex is having an absorbance at
540 nm with molar extinction coefficient of 26,530 L mol−1 cm−1 and 16,990 L mol−1 cm−1 for water and nitric
acid dilution, respectively; the relative standard deviation was found to be <2% and the detection limit was
1 ppm. The interference studies of nitrous acid determination with uranium and zirconium were carried out, and
there is no interference in nitrous acid determination in the range of concentration studied; hence, the optimized
method can be employed for the determination of nitrous acid in plant samples.
Keywords
Plutonium-uranium recovery by extraction Nitrous acid Chromogenic agent Spectrophotometry.References
No references available for this article.
Citation
M.Suba, P.Velavendan, N.K. Pandey, C. Mallika and U. Kamachi Mudali. Spectrophotometric Determination of Nitrous Acid in the Aqueous Streams of Fast Reactor Fuel Reprocessing by using Azo dye Method. Indian J. Adv. Chem. Sci. 2016; 4(3):321-326.