Three New Azido/Thiocyanato Bridged Complexes of CoII/ NiII Derived from Potential Schiff base Blocker Ligand(H2L) of 1-(5-Chloro-2-Hydroxy-Phenyl)-Ethanone and Propane-1,3-diamine-Synthesis, Characterization and Magneto Structural Relationship
Dhrubajyoti Majumdar
DOI:
Volume 4, Issue 3 | Pages: 336-345
Abstract
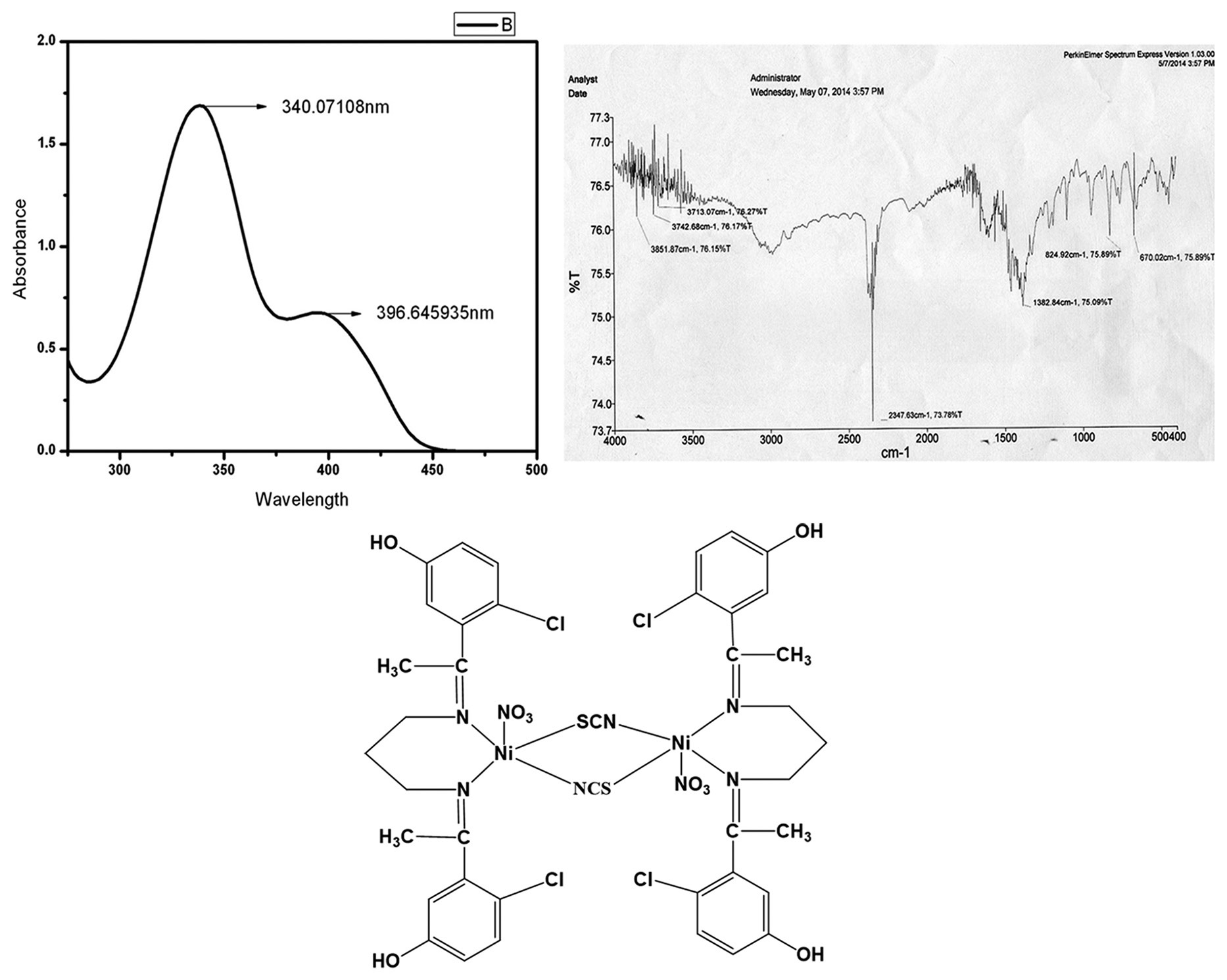
The 2:1 condensation of 1-(5-Chloro-2-hydroxy-phenyl)-ethanone and propane-1,3-diamine gives the tetradentate
azo-linked Schiff base blocker ligand (H2L). The reference Schiff base was allowed to react with methanolic
solution of Co(NO3)2.6H2O and Ni(NO3)2.6H2O separately in the presence of sodium azide/ammonium
thiocyanate yielding formulated complexes (1A), (1B) [Co2(L)2(N3)2](NO3)2, [Co2(L)2(SCN)2](NO3)2 and (2)
[Ni2(H2L)2(SCN)2(NO3)2], respectively. Co(II)/Ni(II) azide/thiocyanate series complexes were systematically
characterized by elemental analysis (C, H and N), molar conductivity study, mass (m/e) and infrared and
ultraviolet-visible (UV-VIS) spectroscopic study. Molar conductivity study unambiguously confirmed that Co(II)
series complexes are completely electrolyte in nature, while Ni(II) complex is nonelectrolyte. The potential Schiff
base blocker ligand (H2L) was further confirmed by1Nuclear magnetic resonance (H NMR) study. Additional
magnetic moment data were provided to establish the expected probable geometry of Co(II), Ni(II) azido/
thiocyanato bridging complexes. All the experimental results suggest that Co(II) bonded to the respective Schiff
base blocker ligand (H2L) through both imino nitrogen and phenolate oxygen along with incorporated azide or
thiocyanate bridging but distinctly imino nitrogen only takes part for Ni(II) complex formation. The electronic
spectra and additional magnetic moment data further confirmed that the environment of Co(II) and Ni(II) metal
ions in both complexes is octahedral or distorted octahedral. Co(II) and Ni(II) series complexes showed magnetic
moment value within the range 4.82-5.08 BM and 2.79 BM, respectively. Literature survey magnetic moment
values of some selected Co(II)/Ni(II) octahedral complexes are good agreement with our synthesized complexes
confirming octahedral geometrical environment around the respective metal ions.
Keywords
Schiff base (H2L) SCN-1 N3-1 bridging Ultraviolet-visible 1H nuclear magnetic resonance Spectra and magnetic moment.References
No references available for this article.
Citation
Dhrubajyoti Majumdar. Three New Azido/Thiocyanato Bridged Complexes of CoII/ NiII Derived from Potential Schiff base Blocker Ligand(H2L) of 1-(5-Chloro-2-Hydroxy-Phenyl)-Ethanone and Propane-1,3-diamine-Synthesis, Characterization and Magneto Structural Relationship. Indian J. Adv. Chem. Sci. 2016; 4(3):336-345.